COVID-19 Vaccines Are Not “Approved” – they are under EUA.
Under section 564 of the Federal Food, Drug, and Cosmetic Act (FD&C Act), when the Secretary of HHS declares that an emergency use authorization is appropriate, FDA may authorize unapproved medical products or unapproved uses of approved medical products to be used in an emergency to diagnose, treat, or prevent serious or life-threatening diseases or conditions caused by CBRN threat agents when certain criteria are met, including there are no adequate, approved, and available alternatives. The HHS declaration to support such use must be based on one of four types of determinations of threats or potential threats by the Secretary of HHS, Homeland Security, or Defense.
What is an Emergency Use Authorization (EUA)?
An Emergency Use Authorization (EUA) is a mechanism to facilitate the availability and use of medical countermeasures, including vaccines, during public health emergencies, such as the current COVID-19 pandemic. Under an EUA, FDA may allow the use of unapproved medical products, or unapproved uses of approved medical products in an emergency to diagnose, treat, or prevent serious or life-threatening diseases or conditions when certain statutory criteria have been met, including that there are no adequate, approved, and available alternatives. Taking into consideration input from the FDA, manufacturers decide whether and when to submit an EUA request to FDA.
Once submitted, FDA will evaluate an EUA request and determine whether the relevant statutory criteria are met, taking into account the totality of the scientific evidence about the vaccine that is available to FDA.
Are the COVID-19 vaccines rigorously tested?
- The FDA says yes.
- Full testing is not complete, which is required for authorization.
- Still under an Emergency Use Authorization.
However, it is likely that it will get full authorization in the future because it has become a purely political objective at this point.
- If COVID-19 vaccines get authorized, that would set a new precedent where previously trials were halted for a handful of deaths.
- According to CDC statistics in VAERS, this vaccine can be temporally (time and place) attributed to >12,000 deaths and >600,000 adverse reactions. This is orders of magnitude above any previous vaccine or even the sum of all vaccines throughout history. Government agencies will deny it but the data is there and the same system has been used throughout history.
Also, the public must not assume the FDA is trustworthy. The government is controlled by big-pharma, which has a revolving door where FDA government employees have lucrative jobs in the big-pharma industry afterwards. Also, the government gives itself legal immunity.
The most important thing is that just because something is approved, it doesn’t mean that you won’t die or get permanently harmed from it. Consider the case of the 1976 Swine Flu government vaccination program.
Safe?
The definition of “safe” is relative. It’s not an absolute value. Most other products are considered unsafe if it causes 1 death. Vaccines can cause many deaths and still be considered safe. Likewise, vaccines can cause permanent injuries such as Guillaine Barre Syndrome and still be considered safe. The government and big-pharma will play games with statistics and clinical correlations but in the end, people get some vaccines and they later either die or get harmed, whether the cause is identified or not. When the government say that “the science doesn’t support that conclusion at this time”, that doesn’t mean the problem doesn’t exist. That just means that they haven’t reproduced it by their procedures. Consider how the federal government once held DDT campaigns and later it was determined to be unsafe.
Regardless of what tests and procedures are in place, nothing changes the fact that people are killed or harmed after vaccinations. Would any of these statistics exist had they not taken the vaccine in the first place? Just look at the statistics.
What happens to you when the government insists the vaccine is safe, but then admits it causes Myocarditis, but then says “too bad, the vaccine is voluntary”. What happens when all the other problems are finally admitted? Does that bring you back to life or fix your permanent injuries?


 March 29, 2025
March 29, 2025
 January 12, 2025
January 12, 2025
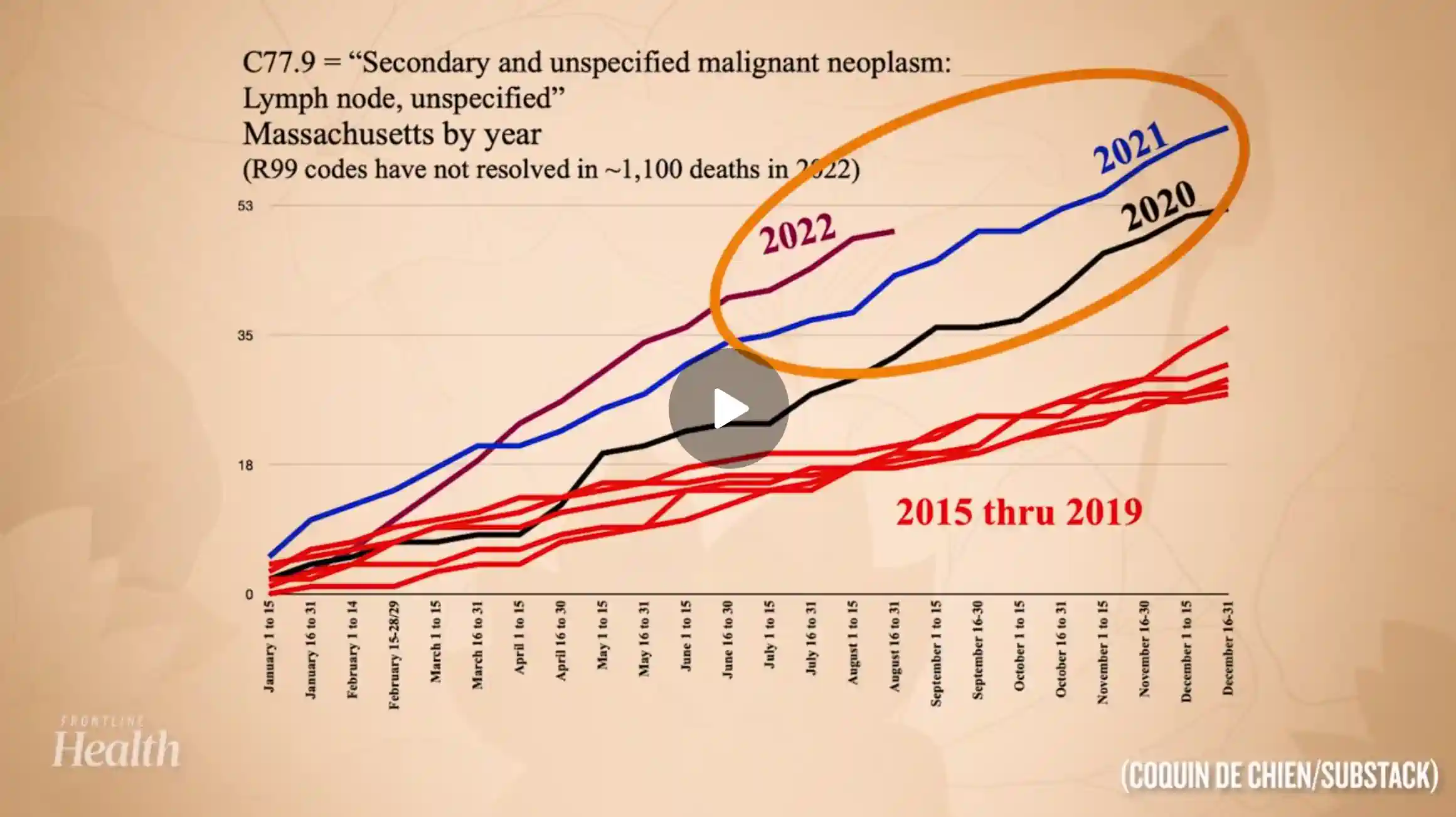 February 6, 2024
February 6, 2024
 February 6, 2024
February 6, 2024
 December 23, 2023
December 23, 2023
 October 28, 2023
October 28, 2023
 June 29, 2023
June 29, 2023
 June 7, 2023
June 7, 2023
 April 23, 2023
April 23, 2023
 March 12, 2023
March 12, 2023
