Executive Summary
Testing is not a reasonable accommodation.
First Principles
- Religious Discrimination
- Only unvaccinated employees are being forced to test because they are exercising their religious rights under the 1st Amendment of the Constitution and Title VII.
- Many people oppose testing because it is a penetration and violation of the temple of the Holy Spirit (1 Corinthians 6 : 19).
- Testing accommodations set precedent for upcoming in-body instant testing where a nano-device is implanted into the body that allows external sensors to gather data from it to confirm either vaccination or illness status. Testing becomes a form of “mark of the beast”.
- Testing is not the least restrictive option.
- Screening is the best option.
- Screening respects privacy, has been used for all previous viral pandemics, and for some professions, is encoded in federal law 14 CFR 121.
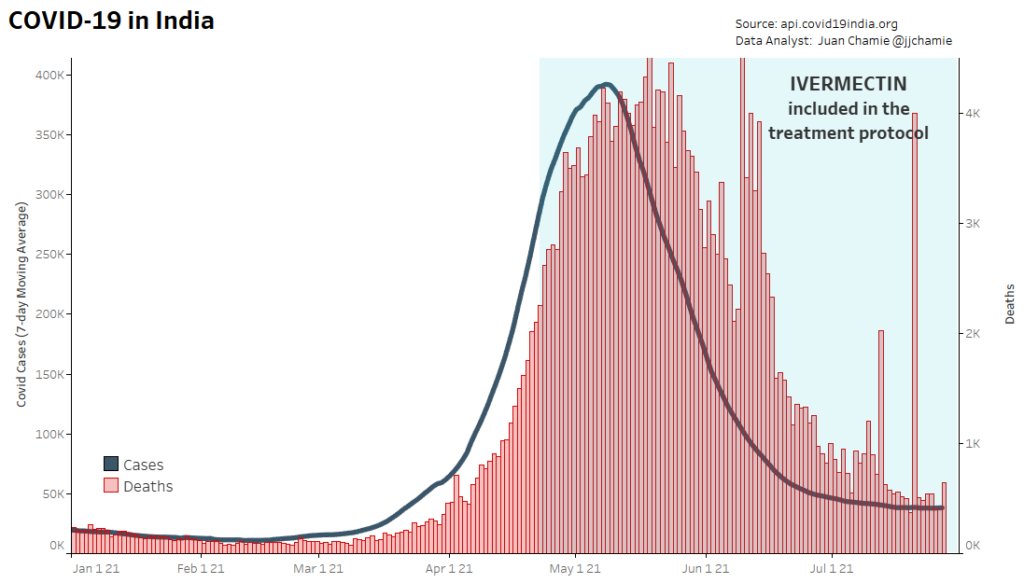
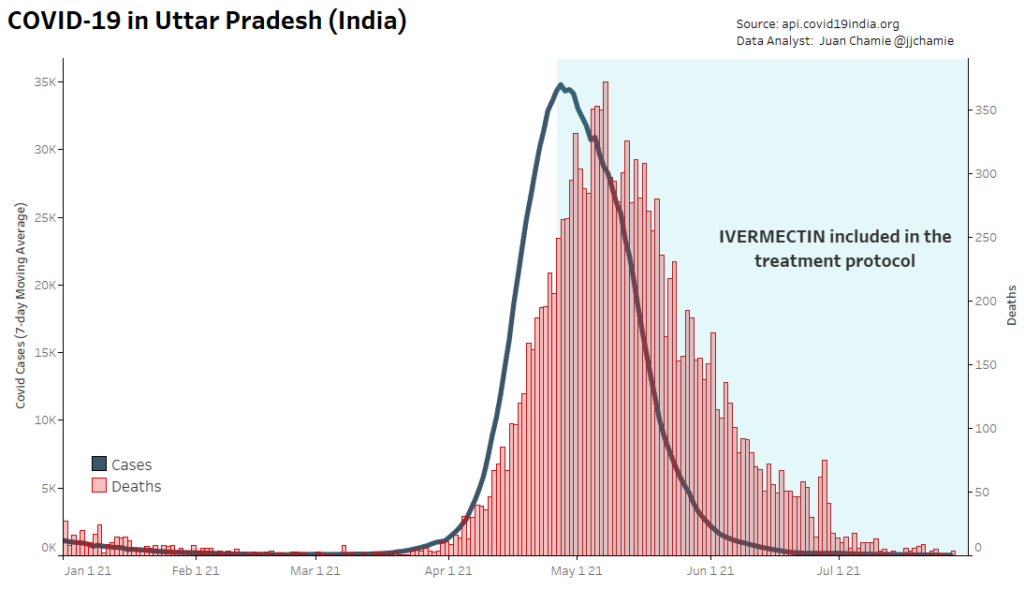
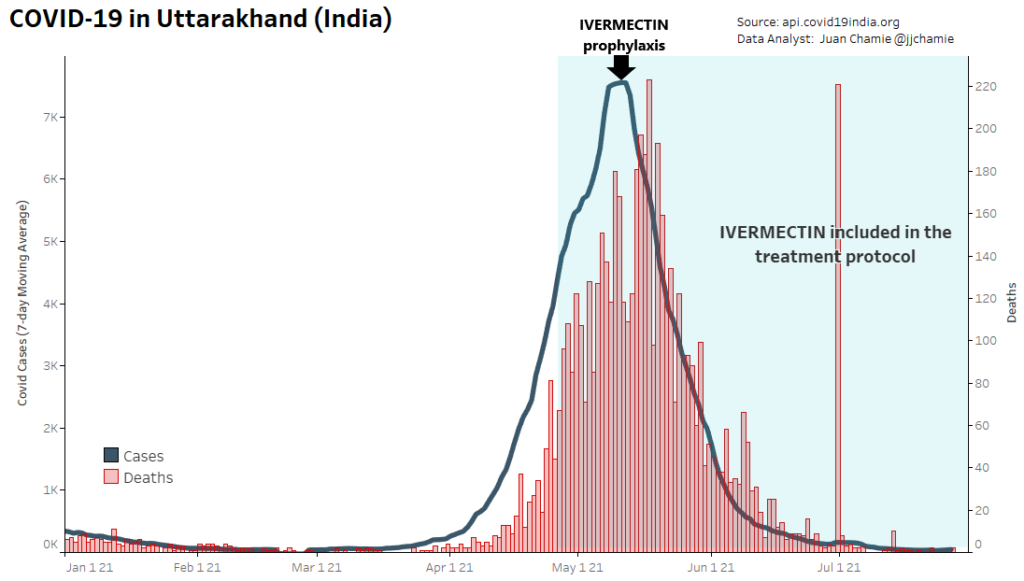
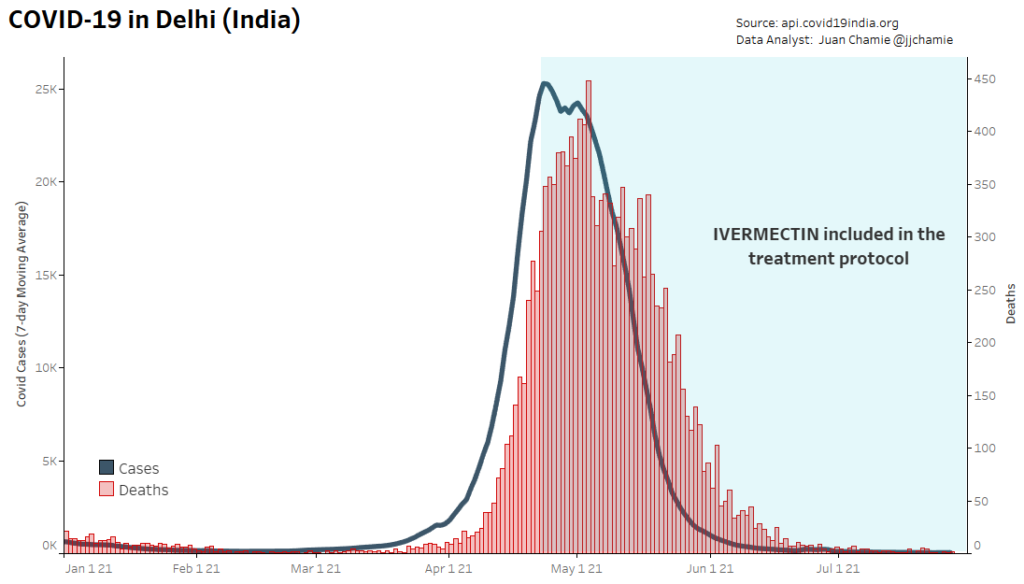
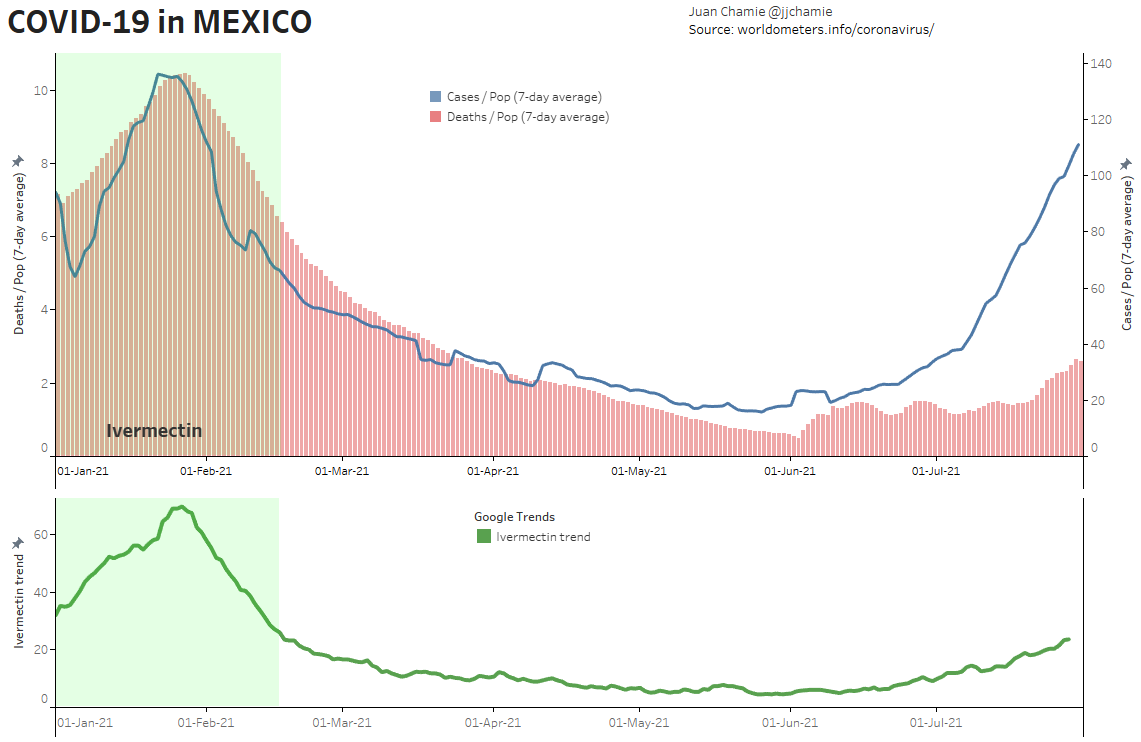
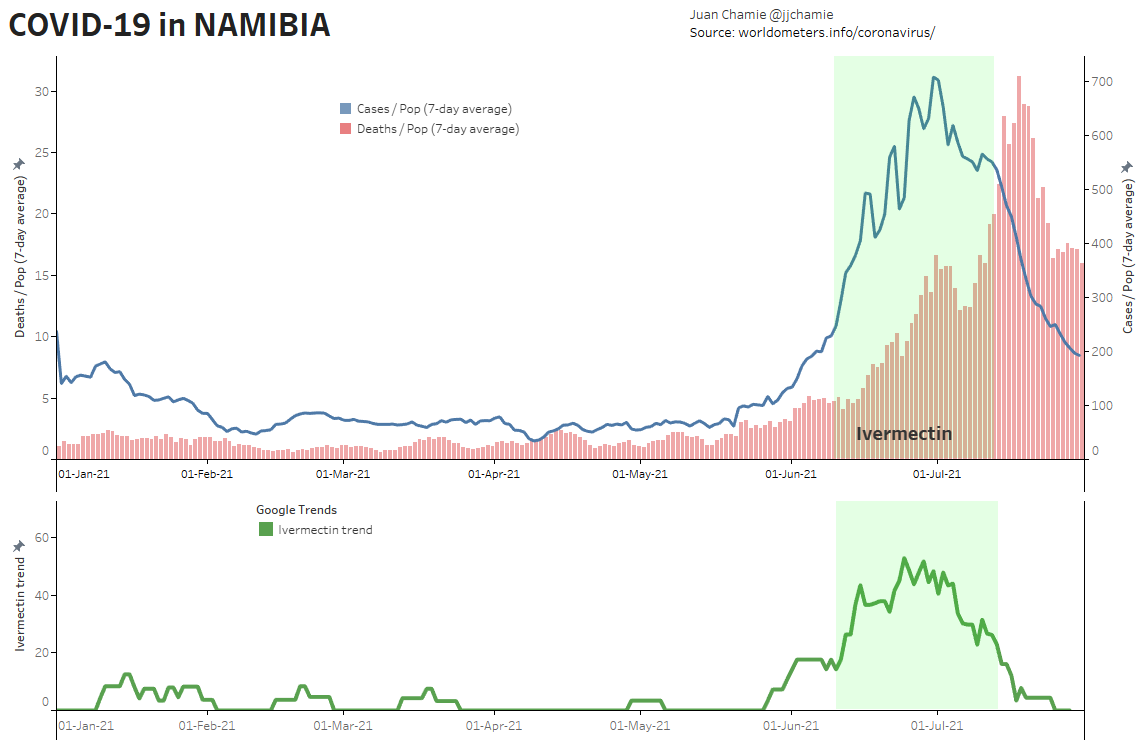
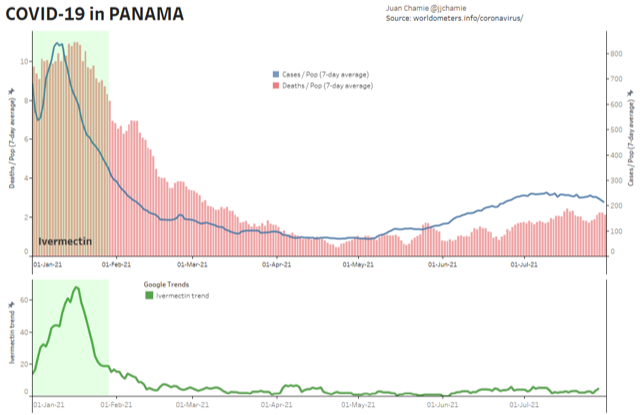
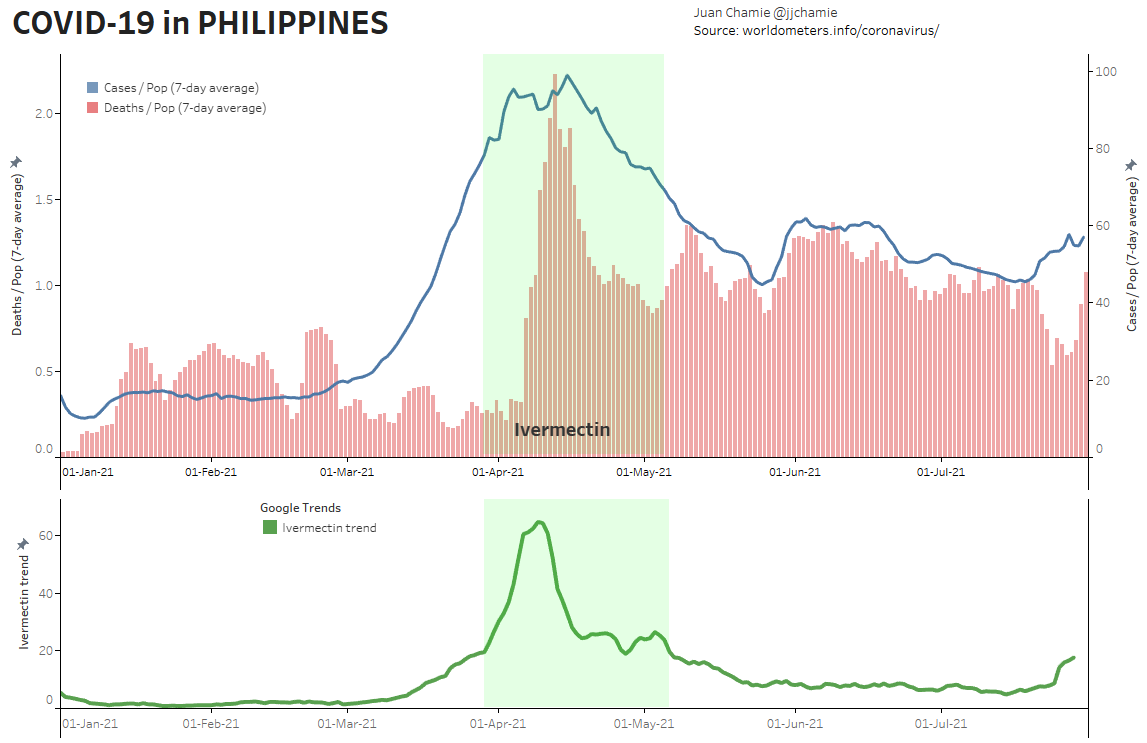
- Prophylactic treatments such as weekly Ivermectin are significantly more effective because they block spike protein binding to ACE2 receptors in cells.
- Social distancing is another option.
- Better sick leave policies will encourage sick people from self-quarantining.
- Financial discrimination.
- Unvaccinated are singled out while equally infectious vaccinated are not required to pay for their own testing, despite vaccines not effective in stopping viral spread.
- Unprecedented.
- Employers do not require weekly screening for HIV/AIDS, Influenza, SARS, MERS, Ebola, etc., despite others being more infectious or more deadly.
Technical Arguments
- Unequal application
- Testing must be required of vaccinated individuals if there is a public health concern because vaccination does not stop infection or spread.
- Vaccines do not stop infection. They are inferior products compared to other products (monoclonal antibodies, repurposed therapeutic drugs, inhaled budesonide, FLCCC protocol).
- Vaccines do not stop spread.
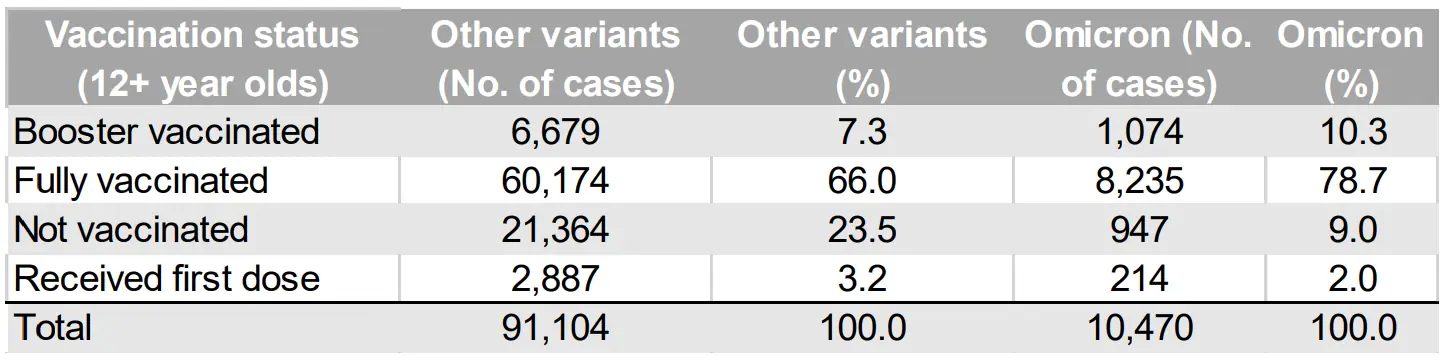
- Denmark study (2021.12.17) demonstrates that vaccines are not effective against the Omicron variant. Therefore, vaccinated people should not be exempted from testing while unvaccinated people are required to be tested. In fact, the opposite is true. It’s not worth testing unvaccinated individuals since they comprise only 9% of the Omicron infection rate. It is the vaccinated who are spreading the Omicron variant.
- 91% of Omicron variant cases are from the combination of fully or partially vaccinated individuals.
- Only 9% of Omicron cases originate from unvaccinated individuals.
- Testing must be required of vaccinated individuals if there is a public health concern because vaccination does not stop infection or spread.
-
- M.D. Anderson cancer patient guidance: “COVID-19 vaccination doesn’t eliminate the need for testing in these medical situations.” source: 11 things to know about COVID-19 testing
- Vaccinated people have the same or higher viral loads meaning that mitigation must be at least equally applied.
- Vaccinated people have less symptoms because of vaccination but they are at least, or more, contagious.
- Vaccinates are traditionally understood to be used to prevent infection and transmission but COVID-19 vaccines have not been tested for either of these. In fact, COVID-19 vaccines have only been placed in clinical trial to reduce symptoms. Therefore, it is non-sensical to discriminate against people exercising religious rights when the vaccinated are at least, or more, infectious than unvaccinated people.
- Testing is non-sensical if the government solution always ends at vaccines. Vaccines are only effective at reducing symptoms and vaccines alone are not a public health strategy since they do not address viral replication and spread. This futile attempt to vaccinate out of a pandemic can be achieved without any testing at all.
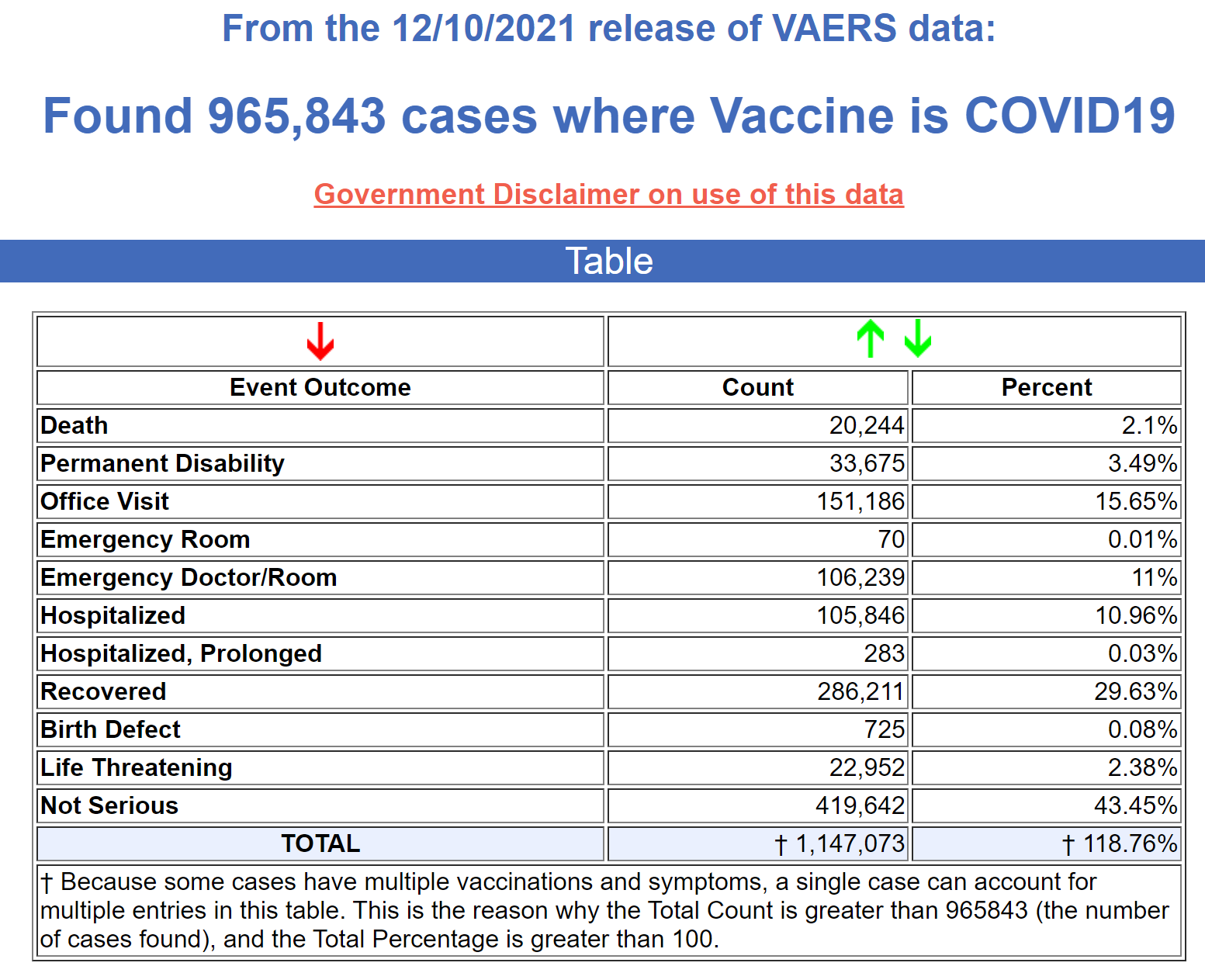
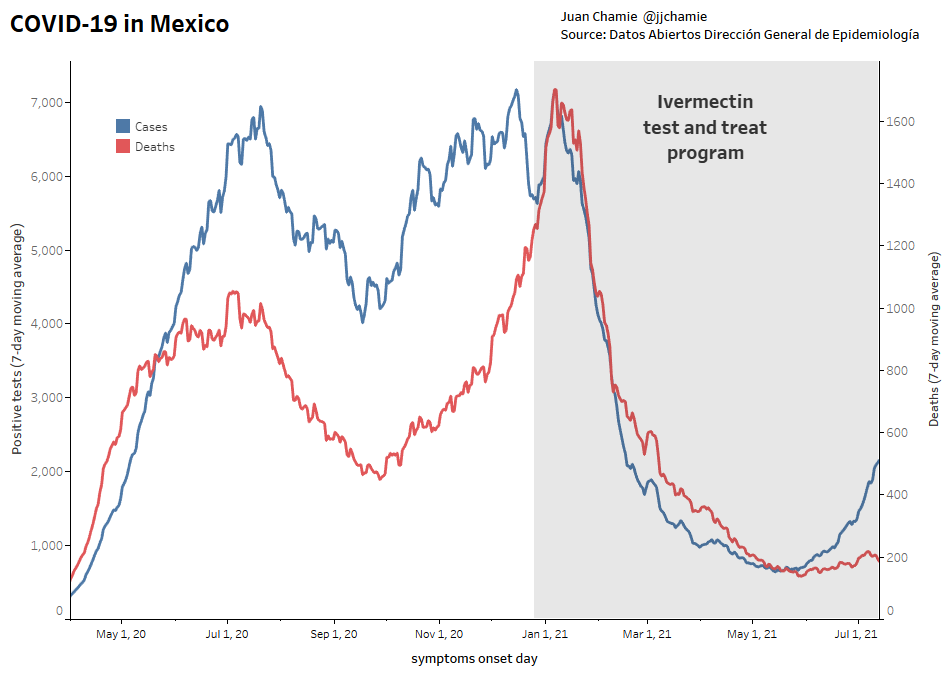
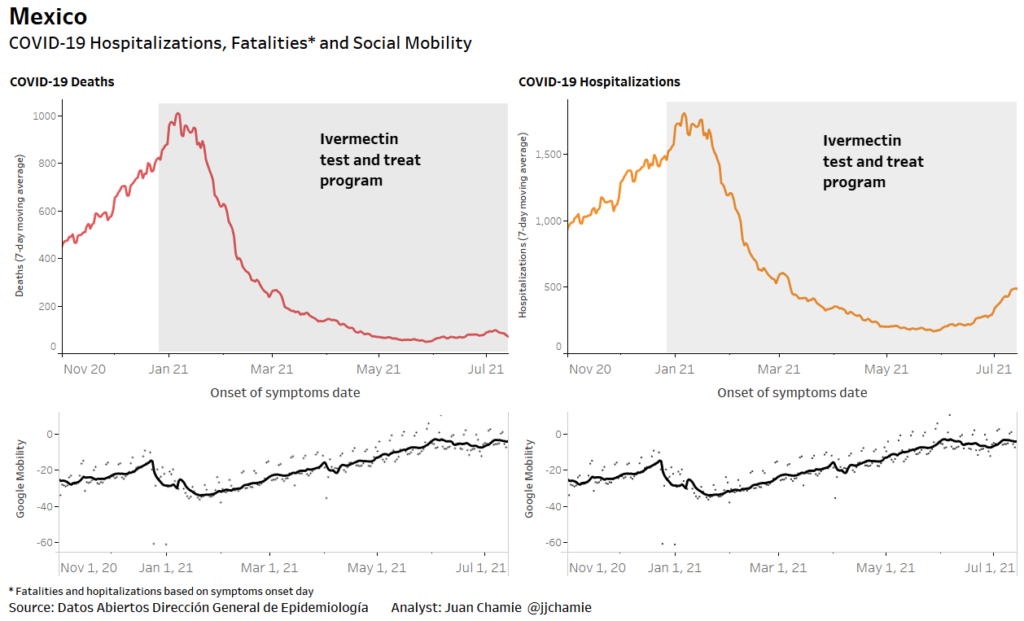
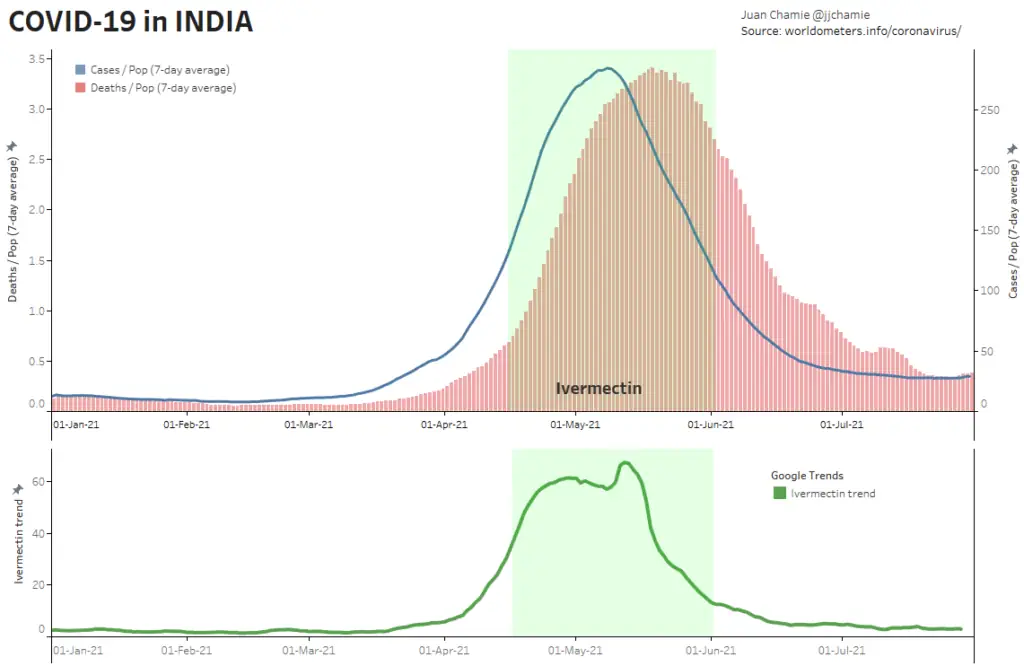
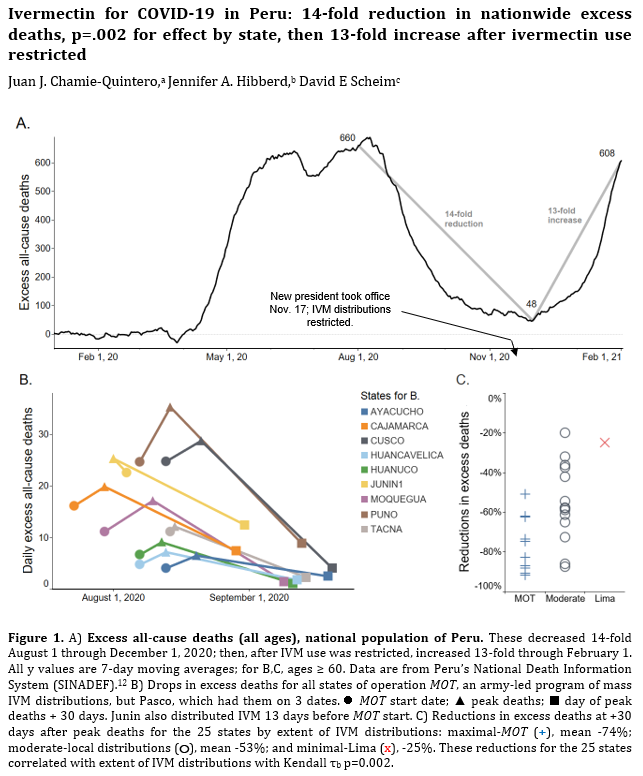
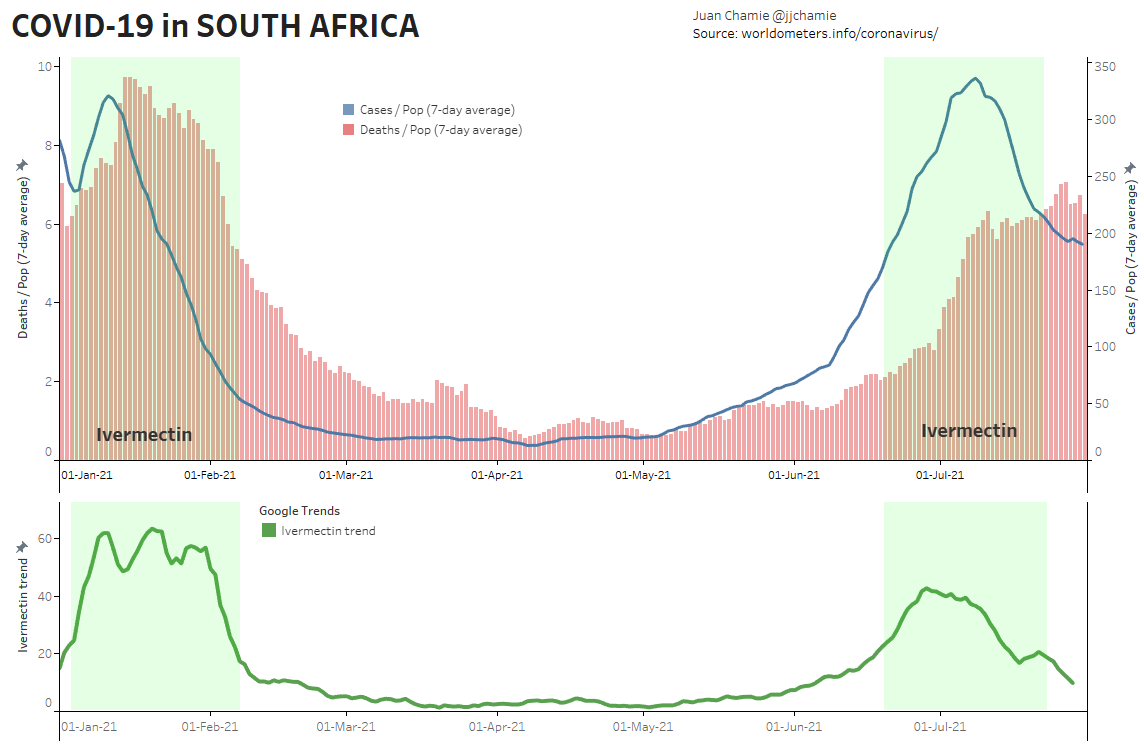
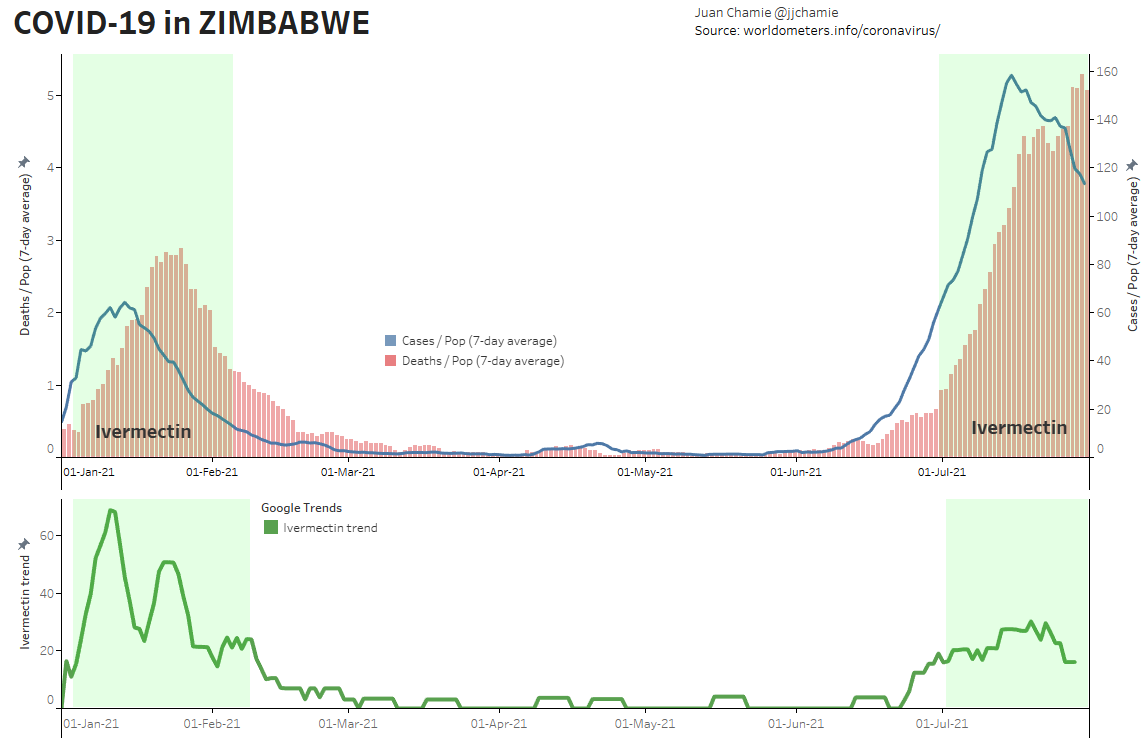
- Alternatively, if public health really were the government concern, the FDA would have issued an EUA for off patent drugs like Ivermectin or inhaled Budesonide, which when used in treatment protocols of repurposed drugs, is significantly more effective based on worldwide data. Reference: The Latest Results of Ivermectin’s Success in Treating Outbreaks of COVID-19
- Spectacular results can be achieved without any testing whatsoever, at a far lower cost, significantly faster timeline, without the substantial risk to life or permanent injury provided by COVID-19 vaccines.
- Alternatively, if public health really were the government concern, the FDA would have issued an EUA for off patent drugs like Ivermectin or inhaled Budesonide, which when used in treatment protocols of repurposed drugs, is significantly more effective based on worldwide data. Reference: The Latest Results of Ivermectin’s Success in Treating Outbreaks of COVID-19
Success Rates of Ivermectin Used in Protocol
- Time between testing and results is too slow to assure public health
- Weekly testing leaves a large period of time in which a person can be infected or be contagious.
- Testing would need to be performed at least before every duty period in order to have any effect.
- Testing performed in other countries, particularly in most highly vaccinated countries, has had no positive effect on reduction of infections.
- Testing is not accurate
- False positives and false negatives occur.
- Testing is part art, part science. Since testing largely uses PCR techniques, it says nothing about viral load, and thus is combined with other techniques such as presentation of symptoms, bronchoscopy, X-ray, or a CT scan of the lungs.
- FDA: (see Policy for Evaluating Impact of Viral Mutations on COVID-19 Tests Guidance for Test Developers and Food and Drug Administration Staff, below)
- “The presence of SARS-CoV-2 mutations in the SARS-CoV-2 virus in a patient sample can potentially change the performance of a test. The clinical impact of mutations on a test’s performance is influenced by various factors”
- “False negative results may occur if mutations are in the part of the viral genome assessed by that test and reduce a test’s ability to detect the virus’s RNA genome.“
- “The impact of genetic variants on molecular test performance is influenced by the sequence of the variant, the design of the test, and the prevalence of the variant in the population. If the test fails to detect a virus variant and the variant increases in prevalence in a community, there may be an increase in the percentage of false negative results.“
- “Changes in the viral genome can result in changes to viral proteins and, therefore, can also impact
the performance of an antigen or serology test.“ - “Antigen tests are designed to detect specific viral proteins. If changes in the viral genome alter the structure of a viral protein targeted by an antigen test, the test may not detect the virus, even if the virus is present, leading to false negative results. The impact of genetic variants on test performance is influenced by the type of change to the protein(s), the design of the test, and the prevalence of the variant in the population.”
- “Serology tests may be designed to detect antibodies produced when the body has an adaptive immune response to an infection. Genetic variants may result in changes to the proteins that elicit an antibody response to the virus and the resulting antibody response. If a serology test is designed to detect antibodies using a particular protein(s), the test may not detect antibodies generated in response to an altered protein configuration. This may result in a false negative test result when antibodies to the virus are present but are not detected by the test. The impact of genetic variants on test performance is influenced by the protein impacted by the genetic change, the type of change to the protein, the resulting change to the antibody response, the design of the test, and the prevalence of the variant in the population.”
- Testing is not an accurate solution: ” As viral mutations can also impact performance of antigen and serology tests, the FDA is also considering the best approach to monitoring the potential effects on EUA-authorized antigen diagnostic and EUA-authorized serology tests.”
- There is a latency between the time a variant emerges and when the variant is detected. Then there is further latency for the FDA to evaluate the effects of the variant against all the tests. These latencies provide a large gap of testing that does not detect the variant. Testing is not automatically sensitive to variants or mutations.
- “…noting that the clinical performance has not been established in all circulating variants, and that performance may vary depending on the variants, and their prevalence, circulating at the time of patient testing.“
- “Recommendations for Developers of Molecular Diagnostic Tests
Since the performance of a diagnostic test can be impacted by viral mutation, FDA recommends that developers: 1) design their test to minimize the impact of viral mutations on test performance; 2) routinely monitor for viral mutations that may impact test performance; and 3) clearly convey any test limitations in the test’s labeling as discussed above.” - “While tests for viral genotyping of new variants may be helpful for monitoring the spread of viral mutations in SARS-CoV-2, it remains unknown whether there is a clinical need for such tests to manage patient care. Tests designed to target and detect specific known variants are likely to become obsolete quickly, as the virus continues to mutate. Therefore, at this time, FDA believes that whole genome sequencing tests may be best suited for genotyping claims due to their ability to detect both known and emerging mutations and variants. FDA recommends that developers of sequencing tests pursuing an EUA with a genotyping claim engage in early discussions with FDA.”
- An inherently accurate test would not need significant refinements. Sometimes multiple types of compound testing are needed to reduce the probability of false positives or negatives.
- “Including a highly conserved pan-SARS-CoV target (a target in a portion of the genetic code associated with the larger Sarbecovirus subgenus of the genus Betacoronavirus in the Coronaviridae family not specific to SARS-CoV-2) as part of a multiple target test may improve performance with a new genetic variant; however, the number of targets in the test should be appropriate to provide resilience (i.e., a reduction of the risk that viral mutation will impact test performance) and most efficiently leverage developer and laboratory resources. When using a highly conserved target in combination with SARS-CoV-2 specific targets, appropriate result interpretation, such as how to interpret results when a pan-SARS-CoV target is positive while the SARS-CoV-2 specific targets are not and follow-up recommendations for an overall positive result with individual gene negative results, may be needed.“
- Testing may be highly inaccurate unless manufacturers constantly monitor their performance, which relies on trust rather than surveillance, leading to the possibility that some tests may be defective and undetected as defective for quite some time.
- “Test developers should assess the potential impact of variants and mutations observed at a significant frequency on their molecular diagnostic test, as well as the potential impact of those viral mutations detected in a smaller percent of the population where there are signs of increasing prevalence. In particular, FDA suggests that developers of molecular tests should periodically conduct sequence alignment of their primer/probe sequences with publicly available SARS-CoV-2 genomes, such as in the GISAID database, to determine the extent to which mutations may impact test performance.“
- Most testing is based on Polymerase Chain Reaction (PCR) testing which is not designed to be a clinical diagnostic tool (reference PCR technique inventor Dr. Kary Mullis).
-
- PCR only amplifies a molecule of a target.
- PCR says nothing about whether the molecule can be cultured so that sufficient amount of viral material exists to actively infect. (reference Anthony Fauci, Dr. Kary Mullis).
- https://rumble.com/embed/vgnrgn/?pub=5zpyl
- FDA results of various tests reflect sensitivity of a particular test vs. the test target but they are still not indicative of culturable viral load. See FDA SARS-CoV-2 Reference Panel Results
- SARS-CoV-2 viral load plateau occurs within a few days of showing symptoms. Testing prior to that provides unreliable results. source: MD Anderson Center
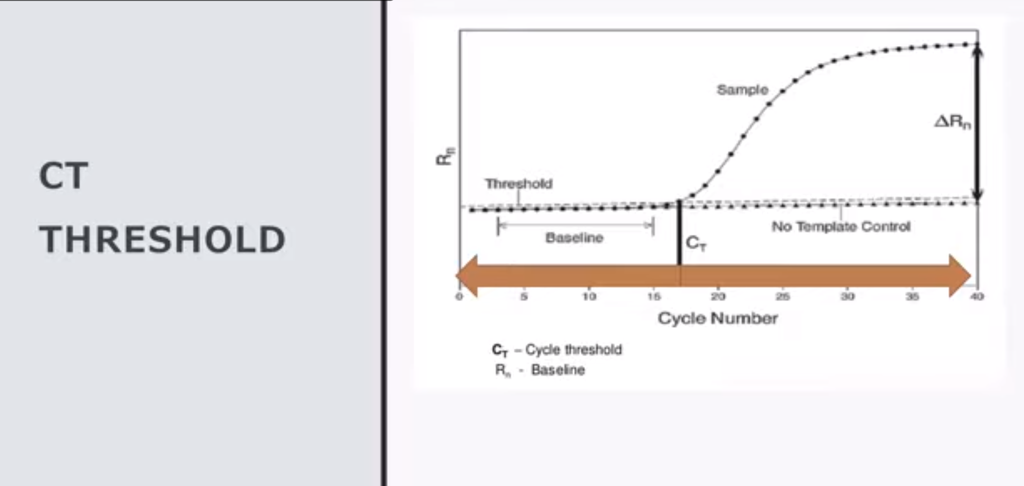
Dr. Anthony Fauci confirms that PCR testing “positives” do not automatically mean that there is sufficient viral load to be replication competent (culturable). In many cases, PCR based testing is picking up dead nucleotides. The USA used cycle thresholds of 40 cycles as a national standard. This has been lowered for vaccinated individuals.
- Testing of asymptomatic people. Experts agree that testing should not be performed on asymptomatic people because of the chance of false positive results.
- There is no evidence of asymptomatic virus spread.
- FDA EUA for PCR based testing expires 2021.12.31 – it cannot distinguish between the Influenza virus and SARS-CoV-2. Reference 07/21/2021: Lab Alert: Changes to CDC RT-PCR for SARS-CoV-2 Testing
- Some tests are unable to detect the Omicron SARS-CoV-2 variant.
- Reference: SARS-CoV-2 Viral Mutations: Impact on COVID-19 Tests
- No privacy of most sacred health information. Requires surrendering human genomic biometric data to third party.
- Intentional and accidental releases will occur giving information to big tech (Microsoft) and the Chinese Communist Party (CCP).
- Contractual obligations will not prevent accidental disclosures, Chinese spy operations, or malevolent behavior. Victims will not be able to prove anything and no degree of compensation will be sufficient to undo harm.
- Some testing is unsafe.
- Testing causes mechanical harm due to proximity to the brain. Testing technicians will cause mechanical harm to some.
- Swab fibers act like asbestos.
- Public health argument is not improved by any activities of the federal government.
- Public health also includes collateral damage (Martin Kulldorff, scientist). For example, the damage caused by a policy which is hypothetical, has proven ineffective around the world, yet targets people exercising their religious rights.
Effects on Delays in Testing
- Dr. Francis Collins, Director of National Institutes of Health, said on NBC’s “Meet the Press” on July 19, 2020, that “[t]he average test delay is too long. And that really undercuts the value of the testing, because you do the testing to find out who’s carrying the virus and then quickly get them isolated so they don’t spread it around. . .We need to do things that are more on the spot. There’s a number of new technologies that are coming along that look very promising in that space. We need to invest a lot of money, and the government is willing to do so, in scaling those up.” NIH director says coronavirus testing delays are ‘too long’ as country tries to control virus
- The Pittsburgh Post-Gazette Editorial Board wrote on July 29, 2020: “Health experts have said from the start of the pandemic testing and contact tracing are keys in controlling the virus, but the delays in obtaining test results make tracing all the more difficult or too late to be effective. Officials estimate if turnaround times extend beyond several days, the results can be useless because the person tested may have already infected scores of people in the interim.”[16]
Types of Tests
Note: sensitive is not the same as accurate.
- Throat swab – less sensitive.
- Saliva – unknown.
- nucleic acid testing, which detects a virus’ genetic material
- antigen testing, which detects the presence of viral proteins that spur the production of antibodies, or the immune system’s response to invaders. Less sensitive than nucleic acid testing. positive antigen test is informative, a negative result would need to be confirmed by the more sensitive nucleic acid test.
M.D. Anderson says COVID nasal swab must be prescribed by a doctor, but this may only be for their brand of testing.
Screening
Screening is an alternative to testing – it is a less restrictive method. It allows the subject to use any means of their choosing to self nominate or disqualify.
- Screening does not create the potential for harm.
- Screening does not violate 1st Amendment religious rights.
- Screening does not violate biometric identity privacy rights.
- Screening can be performed prior to every work cycle, and not just weekly. Weekly testing provides large holes where a person can be infectious but still qualified by testing.
- Screening has been used for decades in safety critical environments (aviation).
- Screening is codified into federal regulations (14 CFR).
Examples of screening:
- 14 CFR 91.17 – aircraft crewmembers are to self-screen for effects of alcohol before every flight.
- 14 CFR § 117.5 – aircrew crewmembers must self-screen for “fitness for duty” from fatigue or any other factor that causes fatigue: medications, lack of sleep, emotional stress, etc.
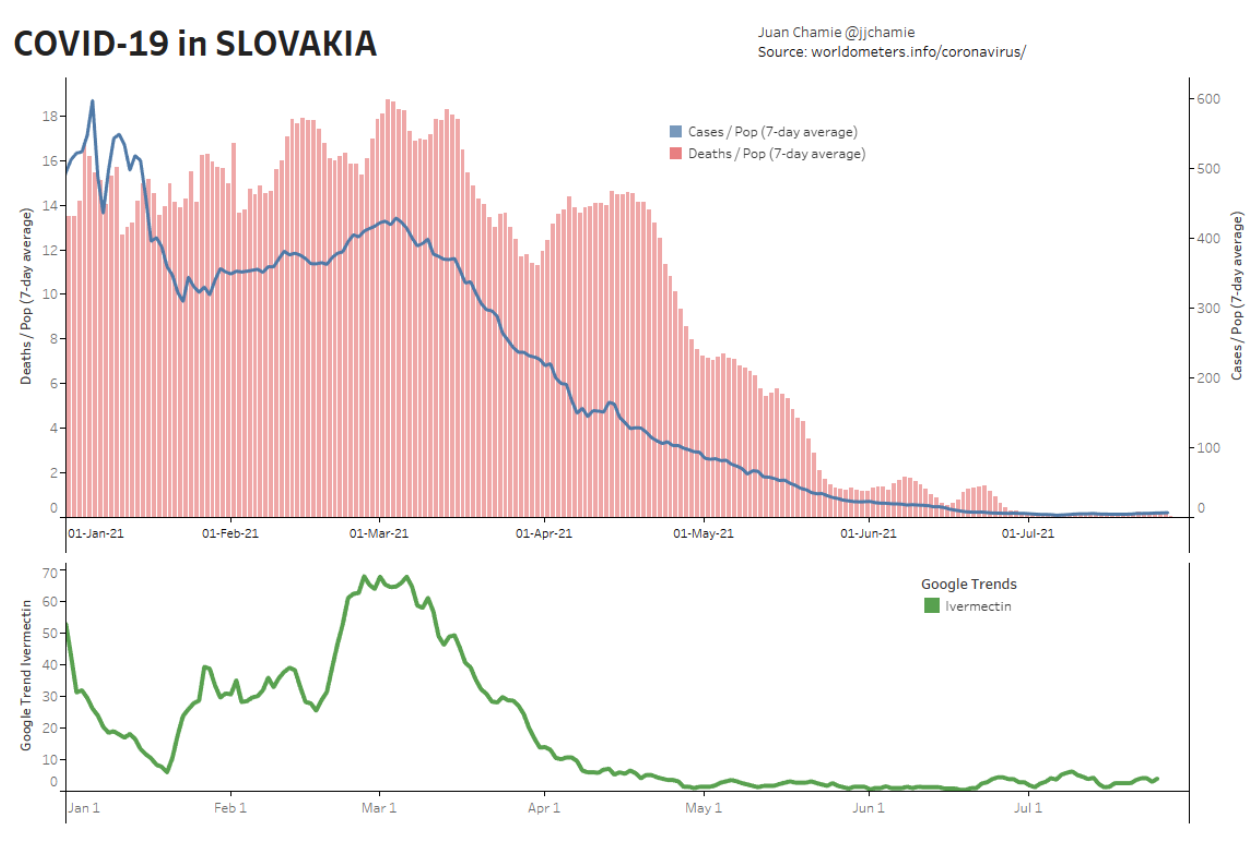
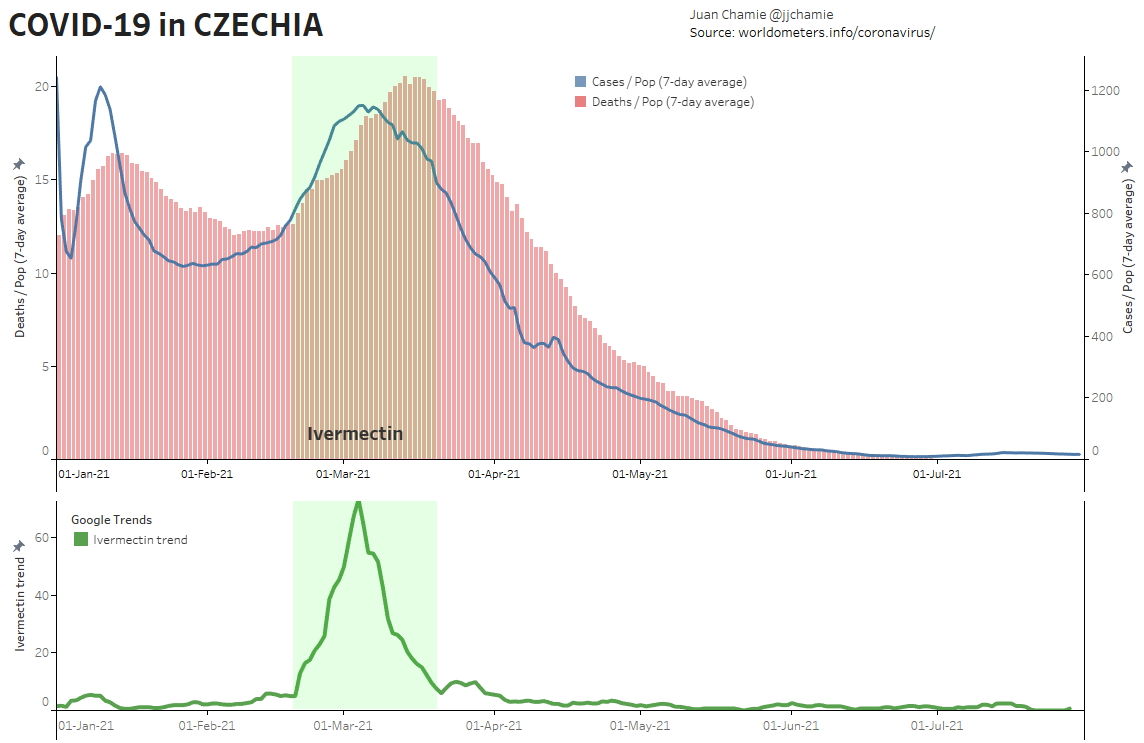
- 14 CFR 382.21 – In aircraft, screening is a practical mitigation method when denial of services could be illegal. This section of federal regulations stipulates what air carriers cannot do unless the passenger condition poses a direct threat. A United Airlines and Department of Defense study demonstrates that aircraft filtration provides sufficient airflow to make transmission risk extremely low.



 June 7, 2023
June 7, 2023
 May 30, 2023
May 30, 2023
 May 21, 2023
May 21, 2023
 November 13, 2022
November 13, 2022
 October 26, 2022
October 26, 2022
 October 20, 2022
October 20, 2022
 September 20, 2022
September 20, 2022
 July 27, 2022
July 27, 2022
 July 12, 2022
July 12, 2022
 May 18, 2022
May 18, 2022