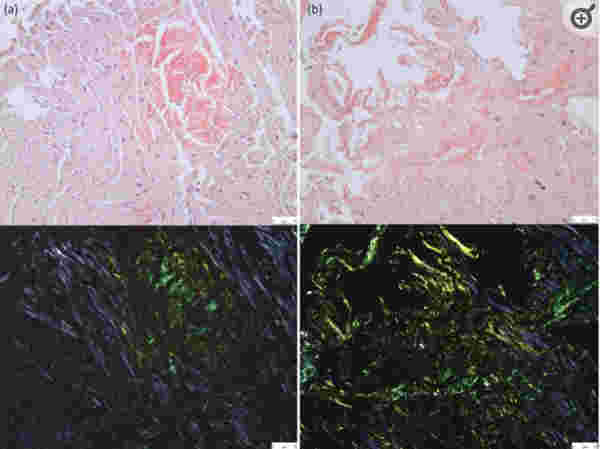
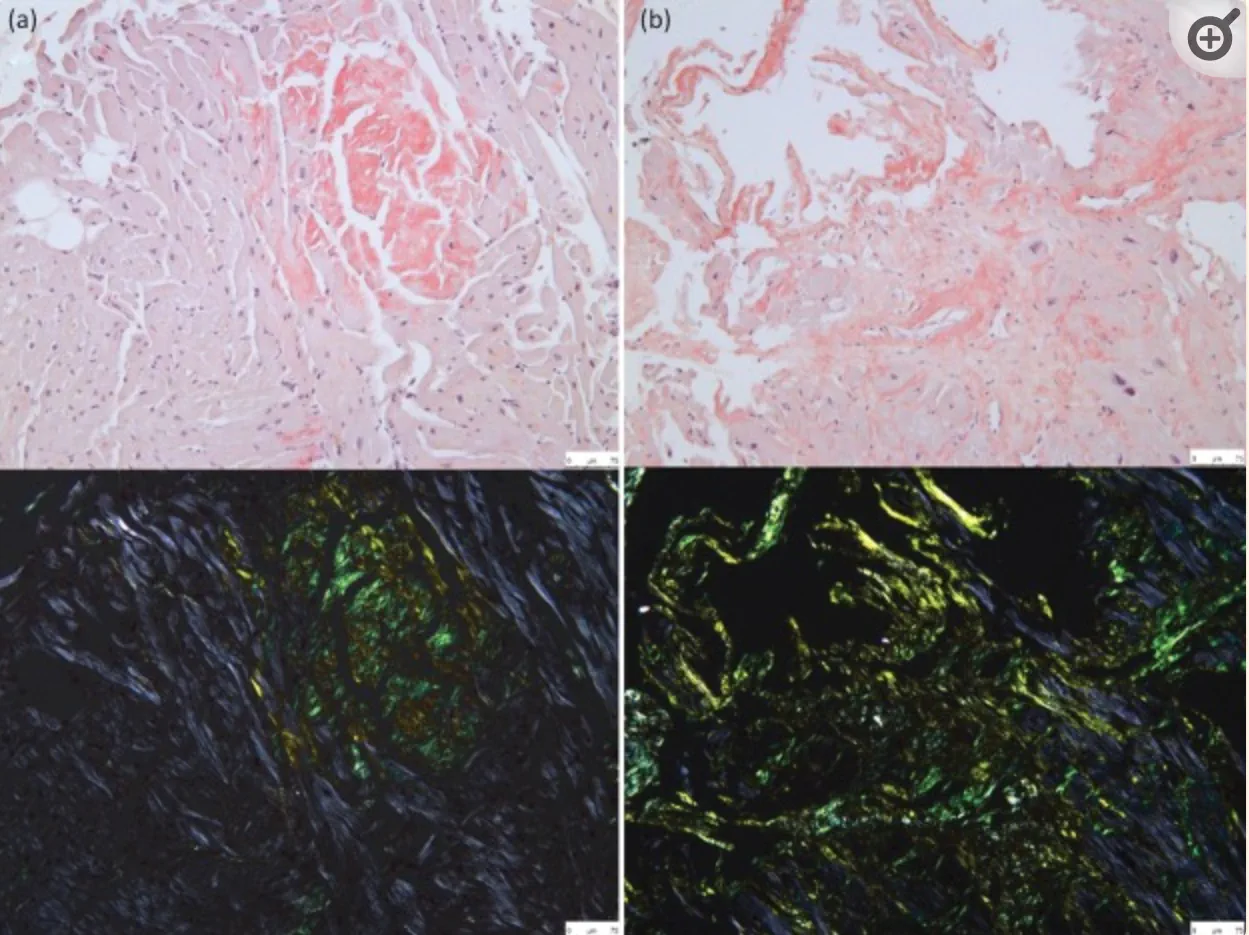
I will be submitting this for peer-reviewed publication when it’s complete.
Although Cardiac Amyloidosis can be inherited, it can also develop from bone marrow (AL – amyloid light chain) or liver (ATTR – amyloid transthyretin) sources. It is more common in men and rare in people under age 40.
A 29-year-old man with no medical history or coronavirus disease 2019 (COVID-19) infection developed chest pain 4 days after his first dose of the Moderna mRNA COVID-19 vaccine (Moderna, MA, USA).
Inflammation of the myocardium (myocarditis) is highly-associated (temporally) with the COVID-19 injectable products.4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24
The figures below are heart tissue specimens stained with Hematoxylin–eosin (H&E) stain which is a stain used for visualizing Eosinophylic amyloid.
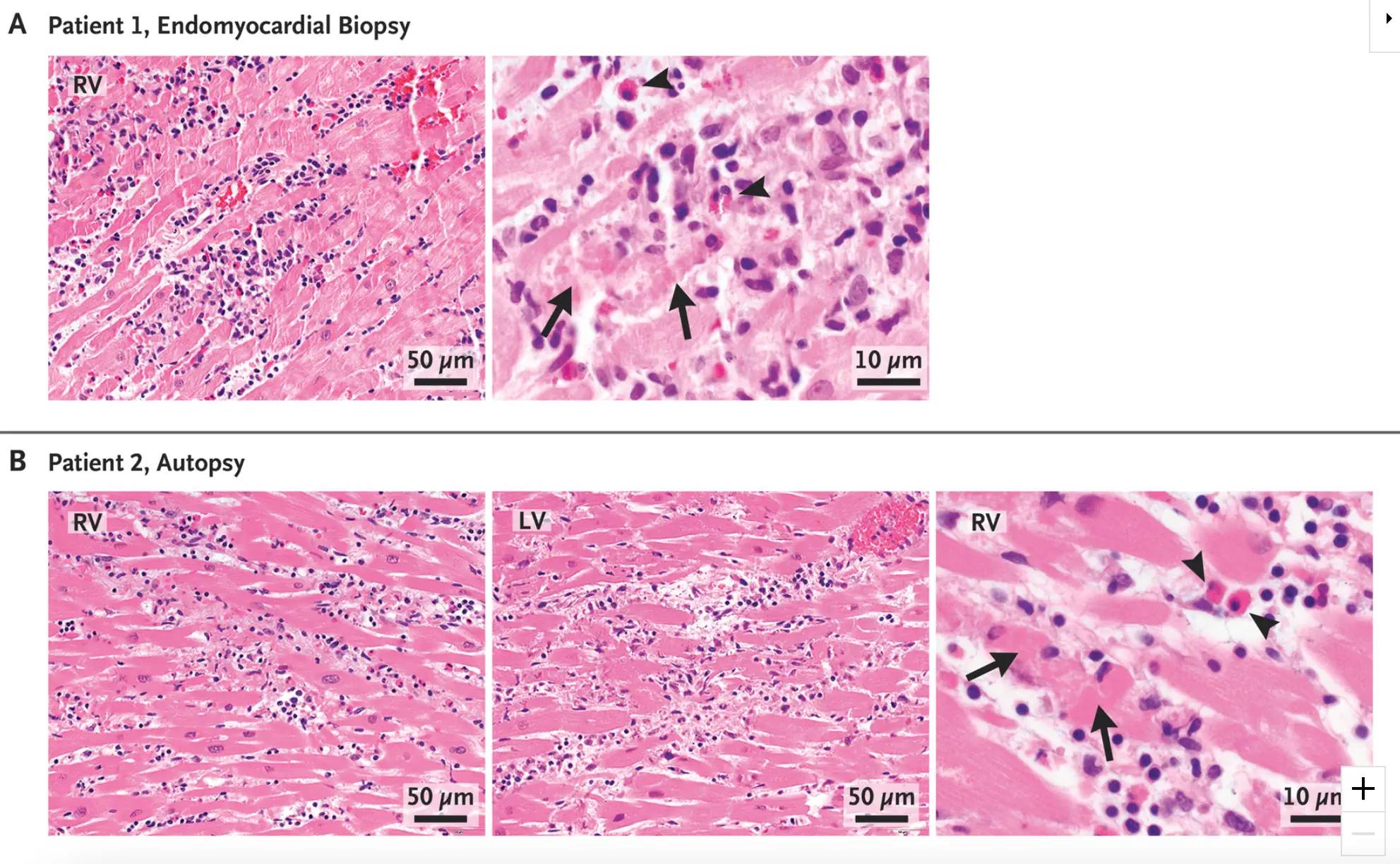
Figure 3: Histopathological Findings from Endomyocardial Biopsy and Autopsy. Hematoxylin–eosin stains of heart-tissue specimens obtained by means of endomyocardial biopsy in patient 1 (Panel A) and autopsy in patient 2 (Panel B) showed myocarditis in both patients, with multifocal cardiomyocyte damage (arrows) associated with mixed inflammatory infiltration. Scattered eosinophils were noted (arrowheads). The images of the hematoxylin–eosin stains were obtained with 10× eyepieces and 40× or 60× objectives. Additional information is provided in the Supplementary Appendix. RV denotes right ventricle, and LV left ventricle. https://pubmed.ncbi.nlm.nih.gov/34407340/
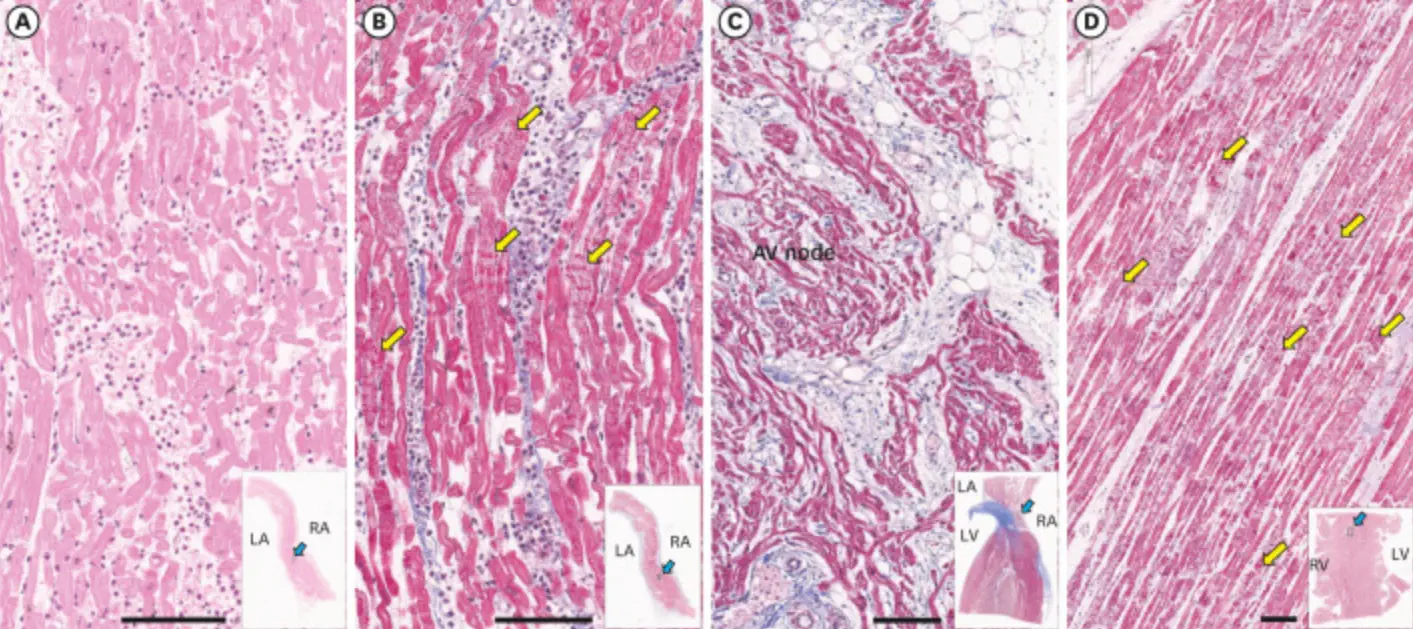
Figure 4: Histopathology of the heart. (A) Hematoxylin and eosin stains of atrial septum shows massive inflammatory infiltration with neutrophil predominance. (B) The myocytes often show contraction band necrosis (yellow arrows), which were highlighted by Masson’s trichrome staining. (C) The atrioventricular node area shows extension of atrial myocarditis to the superficial layer of the node. (D) The ventricular myocardium is free of inflammatory infiltrates, but there are multiple large foci of contraction band necrosis (yellow arrows) particularly in the left ventricular wall and the ventricular septum. Bars represent 100 µm. The blue arrows in insets show where the section was taken from the low magnification views. Hematoxylin and eosin stain was used for the specimen shown in (A) and Masson’s trichrome stain was used for the specimen shown in (B-D). RA = right atrium, LA = left atrium, RV = right ventricle, LV = left ventricle. https://pubmed.ncbi.nlm.nih.gov/34664804/

Figure 5: https://allen.silverchair-cdn.com/allen/content_public/journal/aplm/pap/10.5858_arpa.2021-0435-sa/2/10.5858_arpa.2021-0435-sa.pdf?
Cardiac amyloidosis, also called infiltrating interstitial fibrosis, involves deposition of amyloid in the myocardium. In contrast to traditional myocardial fibrosis, the condition arises independently of cardiac stress or load. In cardiac amyloidosis, ECG and imaging may yield findings that resemble myocardial fibrosis (low voltage, hypertrophy and signs of fibrosis). Various biomarkers and cardiac MRI can distinguish between the conditions. Cardiac amyloidosis causes severe heart failure with a poor prognosis.25
In a study entitled: “Myocarditis is rare in COVID-19 autopsies: cardiovascular findings across 277 postmortem examinations”, 11 cases of Cardiac Amyloidosis were identified in contrast to only 20 cases of myocarditis.
Amyloidosis, albeit not the result of COVID-19, was a more homogeneous finding, being reported in 5 separate studies with the highest incidence in any study being 28.5% (6 of 21 cases). [Halushka MK. et al., 2021]
I shall return.
More references to support this hypothesis.26 “Sudden death in lambda light chain AL cardiac amyloidosis: a review of literature and update for clinicians and pathologists.” Sudden death, eh?
https://www.pennmedicine.org/for-patients-and-visitors/patient-information/conditions-treated-a-to-z/cardiac-amyloidosis
Shah KB, Inoue Y, Mehra MR. Amyloidosis and the Heart: A Comprehensive Review. Arch Intern Med. 2006;166(17):1805–1813. doi:10.1001/archinte.166.17.1805
Martinez-Naharro, A., Hawkins, P. N., & Fontana, M. (2018). Cardiac amyloidosis. Clinical medicine (London, England), 18(Suppl 2), s30–s35. https://doi.org/10.7861/clinmedicine.18-2-s30.
Verma AK, Lavine KJ, Lin CY. Myocarditis after Covid-19 mRNA Vaccination. N Engl J Med. 2021 Sep 30;385(14):1332-1334. doi: 10.1056/NEJMc2109975. Epub 2021 Aug 18. PMID: 34407340; PMCID: PMC8385564.
Choi S, Lee S, Seo JW, Kim MJ, Jeon YH, Park JH, Lee JK, Yeo NS. Myocarditis-induced Sudden Death after BNT162b2 mRNA COVID-19 Vaccination in Korea: Case Report Focusing on Histopathological Findings. J Korean Med Sci. 2021 Oct 18;36(40):e286. doi: 10.3346/jkms.2021.36.e286. PMID: 34664804; PMCID: PMC8524235.
Sessa F, Salerno M, Esposito M, Di Nunno N, Zamboni P, Pomara C. Autopsy Findings and Causality Relationship between Death and COVID-19 Vaccination: A Systematic Review. J Clin Med. 2021 Dec 15;10(24):5876. doi: 10.3390/jcm10245876. PMID: 34945172; PMCID: PMC8709364.
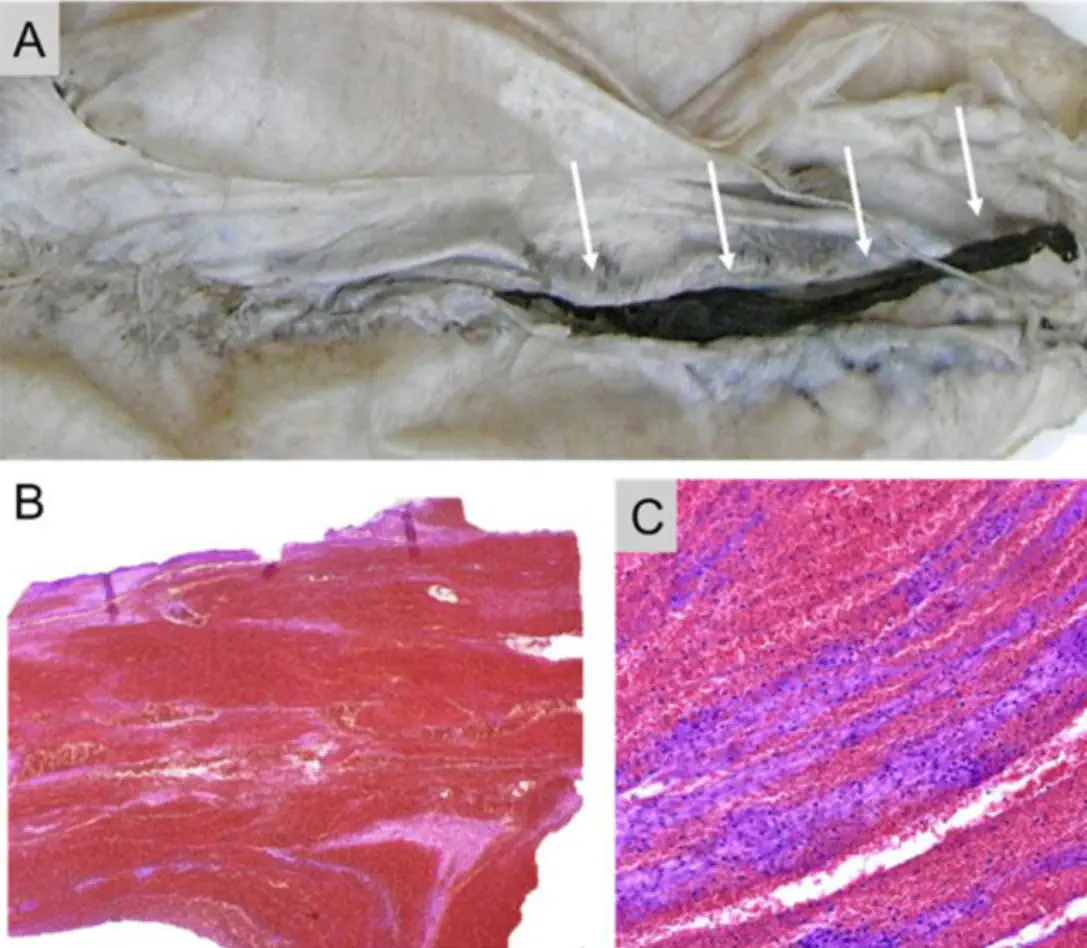
Gill JR, Tashjian R, Duncanson E. Autopsy Histopathologic Cardiac Findings in Two Adolescents Following the Second COVID-19 Vaccine Dose. Arch Pathol Lab Med. 2022 Feb 14. doi: 10.5858/arpa.2021-0435-SA. Epub ahead of print. PMID: 35157759.
Barton LM, Duval EJ, Stroberg E, Ghosh S, Mukhopadhyay S. COVID-19 Autopsies, Oklahoma, USA. Am J Clin Pathol. 2020 May 5;153(6):725-733. doi: 10.1093/ajcp/aqaa062. Erratum in: Am J Clin Pathol. 2020 May 5;153(6):852. PMID: 32275742; PMCID: PMC7184436.
Ilonze OJ, Guglin ME. Myocarditis following COVID-19 vaccination in adolescents and adults: a cumulative experience of 2021. Heart Fail Rev. 2022 Apr 22:1–11. doi: 10.1007/s10741-022-10243-9. Epub ahead of print. PMID: 35449353; PMCID: PMC9023259.
Truong DT et al. Clinically Suspected Myocarditis Temporally Related to COVID-19 Vaccination in Adolescents and Young Adults: Suspected Myocarditis After COVID-19 Vaccination. Circulation. 2022 Feb;145(5):345-356. doi: 10.1161/CIRCULATION AHA.121.056583. Epub 2021 Dec 6. PMID: 34865500.
Dionne A, Sperotto F, Chamberlain S, Baker AL, Powell AJ, Prakash A, Castellanos DA, Saleeb SF, de Ferranti SD, Newburger JW, Friedman KG. Association of Myocarditis With BNT162b2 Messenger RNA COVID-19 Vaccine in a Case Series of Children. JAMA Cardiol. 2021 Dec 1;6(12):1446-1450. doi: 10.1001/jamacardio.2021.3471. PMID: 34374740; PMCID: PMC8356143.
Kim HW, Jenista ER, Wendell DC, Azevedo CF, Campbell MJ, Darty SN, Parker MA, Kim RJ. Patients With Acute Myocarditis Following mRNA COVID-19 Vaccination. JAMA Cardiol. 2021 Oct 1;6(10):1196-1201. doi: 10.1001/jamacardio.2021.2828. PMID: 34185046; PMCID: PMC8243258.
Navar AM, McNally E, Yancy CW, O’Gara PT, Bonow RO. Temporal Associations Between Immunization With the COVID-19 mRNA Vaccines and Myocarditis: The Vaccine Safety Surveillance System Is Working. JAMA Cardiol. 2021 Oct 1;6(10):1117-1118. doi: 10.1001/jamacardio.2021.2853. PMID: 34185044.
Shay DK, Shimabukuro TT, DeStefano F. Myocarditis Occurring After Immunization With mRNA-Based COVID-19 Vaccines. JAMA Cardiol. 2021 Oct 1;6(10):1115-1117. doi: 10.1001/jamacardio.2021.2821. PMID: 34185047.
Montgomery J, Ryan M, Engler R, Hoffman D, McClenathan B, Collins L, Loran D, Hrncir D, Herring K, Platzer M, Adams N, Sanou A, Cooper LT Jr. Myocarditis Following Immunization With mRNA COVID-19 Vaccines in Members of the US Military. JAMA Cardiol. 2021 Oct 1;6(10):1202-1206. doi: 10.1001/jamacardio.2021.2833. PMID: 34185045; PMCID: PMC8243257.
Bozkurt B, Kamat I, Hotez PJ. Myocarditis With COVID-19 mRNA Vaccines. Circulation. 2021 Aug 10;144(6):471-484. doi: 10.1161/CIRCULATIONAHA.121.056135. Epub 2021 Jul 20. PMID: 34281357; PMCID: PMC8340726.
Simone A, Herald J, Chen A, Gulati N, Shen AY, Lewin B, Lee MS. Acute Myocarditis Following COVID-19 mRNA Vaccination in Adults Aged 18 Years or Older. JAMA Intern Med. 2021 Dec 1;181(12):1668-1670. doi: 10.1001/jamainternmed.2021.5511. PMID: 34605853; PMCID: PMC8491129.
Maeda M, Isawa T, Tada N. Definite Acute Myocarditis After Coronavirus Disease 2019 mRNA Vaccination. Circ J. 2022 Mar 25;86(4):724. doi: 10.1253/circj.CJ-21-0840. Epub 2021 Dec 4. PMID: 34866122.
Schneider J, Sottmann L, Greinacher A, Hagen M, Kasper HU, Kuhnen C, Schlepper S, Schmidt S, Schulz R, Thiele T, Thomas C, Schmeling A. Postmortem investigation of fatalities following vaccination with COVID-19 vaccines. Int J Legal Med. 2021 Nov;135(6):2335-2345. doi: 10.1007/s00414-021-02706-9. Epub 2021 Sep 30. PMID: 34591186; PMCID: PMC8482743.
Cushion S, Arboleda V, Hasanain Y, Demory Beckler M, Hardigan P, Kesselman MM. Comorbidities and Symptomatology of SARS-CoV-2 (Severe Acute Respiratory Syndrome Coronavirus 2)-Related Myocarditis and SARS-CoV-2 Vaccine-Related Myocarditis: A Review. Cureus. 2022 Apr 12;14(4):e24084. doi: 10.7759/cureus.24084. PMID: 35573496; PMCID: PMC9099161.
Kang DH, Na JY, Yang JH, Moon SH, Kim SH, Jung JJ, Cha HJ, Ahn JH, Park YW, Cho SY, Yu HK, Lee SH, Park MY, Kim JW, Byun JH. Fulminant Giant Cell Myocarditis following Heterologous Vaccination of ChAdOx1 nCoV-19 and Pfizer-BioNTech COVID-19. Medicina (Kaunas). 2022 Mar 20;58(3):449. doi: 10.3390/medicina58030449. PMID: 35334625; PMCID: PMC8950462.
Kim SH, Lee SY, Kim GY, Oh JS, Kim J, Chun KJ, Ju MH, Lee CH, Song YJ, Na JY. A Case of Heart Transplantation for Fulminant Myocarditis After ChAdOx1 nCoV-19 Vaccination. J Korean Med Sci. 2022 Apr 4;37(13):e104. doi: 10.3346/jkms.2022.37.e104. PMID: 35380028; PMCID: PMC8980363.
Kornowski R, Witberg G. Acute myocarditis caused by COVID-19 disease and following COVID-19 vaccination. Open Heart. 2022 Mar;9(1):e001957. doi: 10.1136/openhrt-2021-001957. PMID: 35264415; PMCID: PMC8914394.
Halushka MK, Vander Heide RS. Myocarditis is rare in COVID-19 autopsies: cardiovascular findings across 277 postmortem examinations. Cardiovasc Pathol. 2021 Jan-Feb;50:107300. doi: 10.1016/j.carpath.2020.107300. Epub 2020 Oct 23. PMID: 33132119; PMCID: PMC7583586.
https://tidsskriftet.no/en/2018/10/oversiktsartikkel/myocardial-fibrosis
D’Errico S, Mazzanti A, Baldari B, Maiese A, Frati P, Fineschi V. Sudden death in lambda light chain AL cardiac amyloidosis: a review of literature and update for clinicians and pathologists. Int J Clin Exp Pathol. 2020;13(7):1474-1482. Published 2020 Jul 1.