Two weeks ago, the FDA begged a Texas judge to delay production on the first monthly batch of 55,000 pages of Covid-19 vaccine data submitted to the agency by Pfizer. Originally, the agency was set to produce just 500 pages-per-month.
Now, Pfizer – which just forecast $54 billion in Covid-related sales in 2022, appears to be anticipating some bad news, as evidenced by several redline changes in their Q4 earnings releases.
As Rubicon Capital’s Kelly Brown notes on Twitter, the changes center around disclosures of unfavorable safety data.
For example, in Q4 they added: “or further information regarding the quality of pre-clinical, clinical or safety data, including by audit or inspection.”

More from Brown, who notes that Pfizer is now highlighting “concerns about clinical data integrity…”

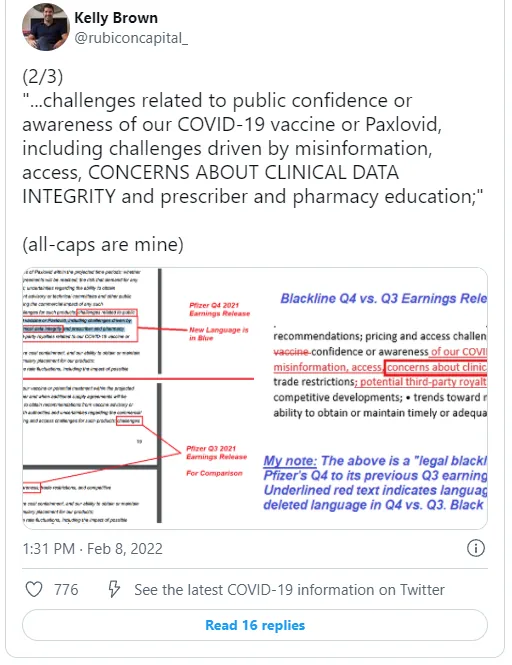
(2/3)
“…challenges related to public confidence or awareness of our COVID-19 vaccine or Paxlovid, including challenges driven by misinformation, access, CONCERNS ABOUT CLINICAL DATA INTEGRITY and prescriber and pharmacy education;”
(all-caps are mine) pic.twitter.com/bAKH4iqiKs
— Kelly Brown (@rubiconcapital_) February 8, 2022
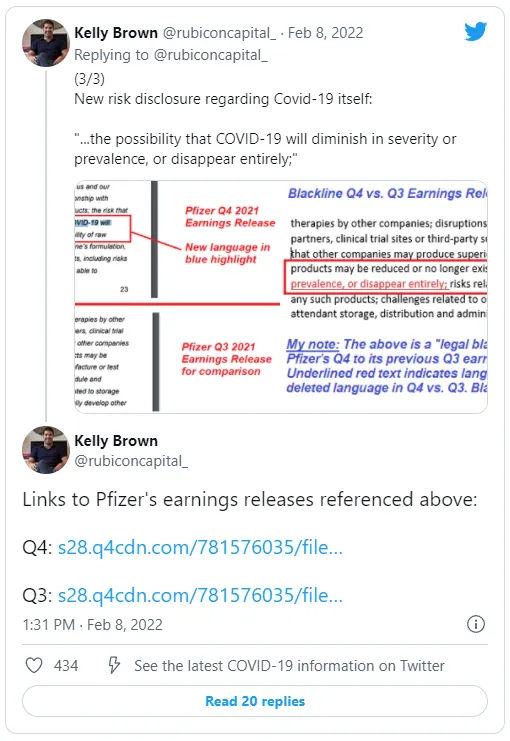
The company also notes that Covid-19 may “diminish in severity or prevalence, or disappear entirely.”
Links to Pfizer’s earnings releases referenced above:
Q4: https://t.co/yCd1Ga8gPu
Q3: https://t.co/CJ8k9oGQ1U
— Kelly Brown (@rubiconcapital_) February 8, 2022
What’s behind the curtain, Pfizer?
Content retrieved from: https://www.zerohedge.com/covid-19/pfizer-quietly-adds-language-warning-unfavorable-pre-clinical-clinical-or-safety-data-may.

