Original article: https://www.medrxiv.org/content/10.1101/2021.07.10.21260232v2 (SARS-CoV-2 variants of concern exhibit reduced sensitivity to live-virus neutralization in sera from CoronaVac vaccinees and naturally infected COVID-19 patients)
PDF of article
Excerpt:
Abstract
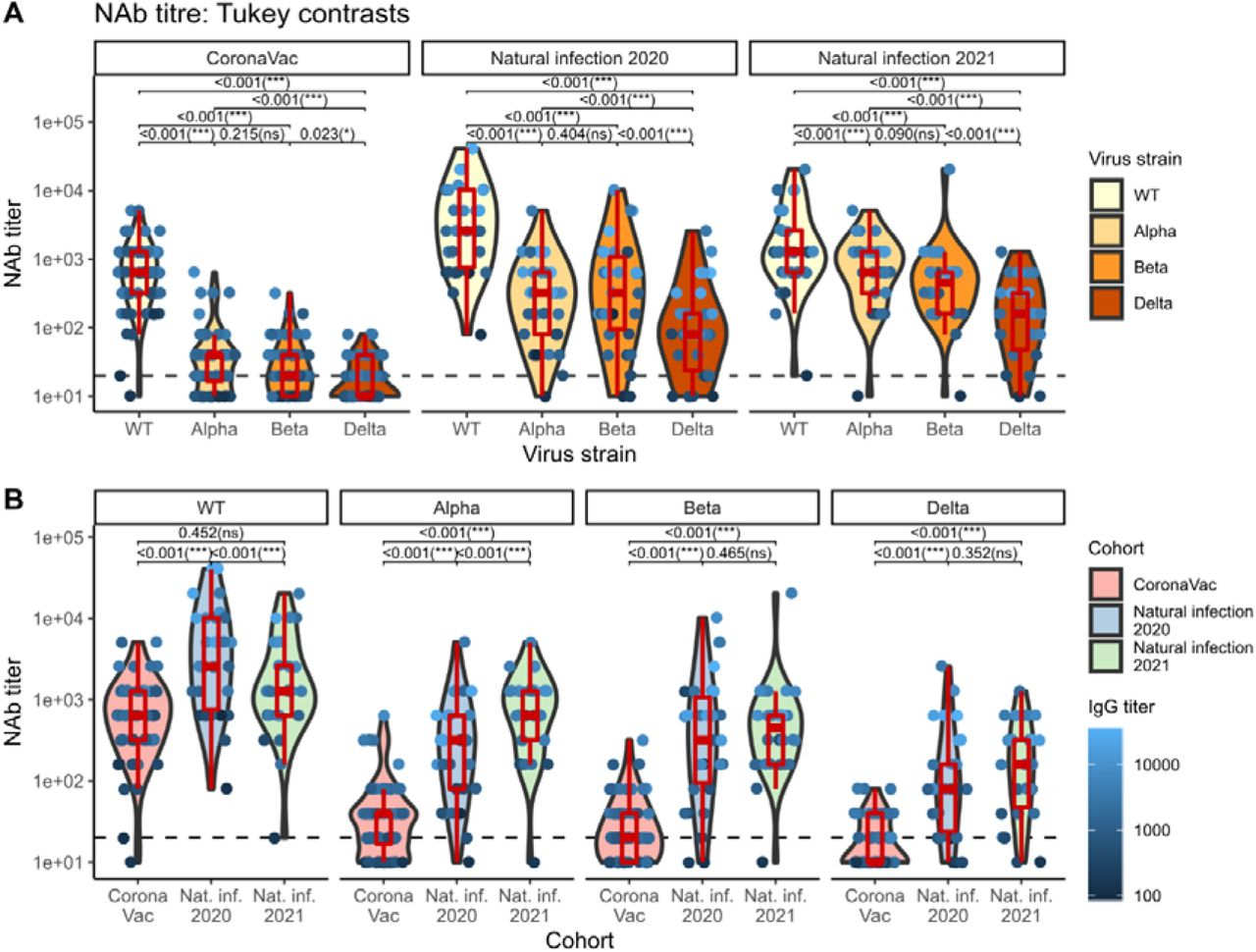
Recent surges in SARS-CoV-2 variants of concern (VOCs) call for the need to evaluate levels of vaccine-and infection-induced SARS-CoV-2 neutralizing antibodies (NAbs). CoronaVac (Sinovac Biotech, Beijing, China) is currently being used for mass vaccination in Thailand as well as other low-income countries. Three VOCs currently circulating within Thailand include the B.1.1.7 (Alpha), B.1.351 (Beta), and B.1.617.2 (Delta) strains. We assessed NAb potency against the prototypic strain containing the original spike sequence (WT) compared to that against the 3 VOCs using sera derived from a cohort of healthcare workers who received a full 2-dose regimen of CoronaVac. Sera from two other cohorts consisting of COVID-19 patients who had been hospitalized in 2020 and 2021 were evaluated for comparison. We found that, despite equally robust production of S1-RBD-binding IgG and 100% seropositivity, sera from both CoronaVac vaccinees and naturally infected individuals had significantly reduced neutralizing capacity against all 3 VOCs compared to WT. Strikingly, NAb titers against Alpha and Beta were comparable, but Delta appears to be significantly more refractory to NAbs in all groups. Our results may help inform on CoronaVac NAb-inducing capacity, which is a proxy for vaccine efficacy, in the context of the WT strain and 3 VOCs. Our results also have critical implications for public health decision-makers who may need to maintain efficient mitigation strategies amid a potentially high risk for infection with VOCs even in those who have been previously infected.
CC-BY-NC-ND 4.0 International license.