Proving yet again, that once you get locked into the vaccine rona-coaster, you are not getting off until the ride’s over. The CDC has recommended, and the FDA has approved COVID-19 booster shots number four and/or number five depending on your life expectancy and vulnerability status.
 Step right up comrades and get em’ fast, while there’s still time before Big Pharma first quarter financial statements are closed.
Step right up comrades and get em’ fast, while there’s still time before Big Pharma first quarter financial statements are closed.
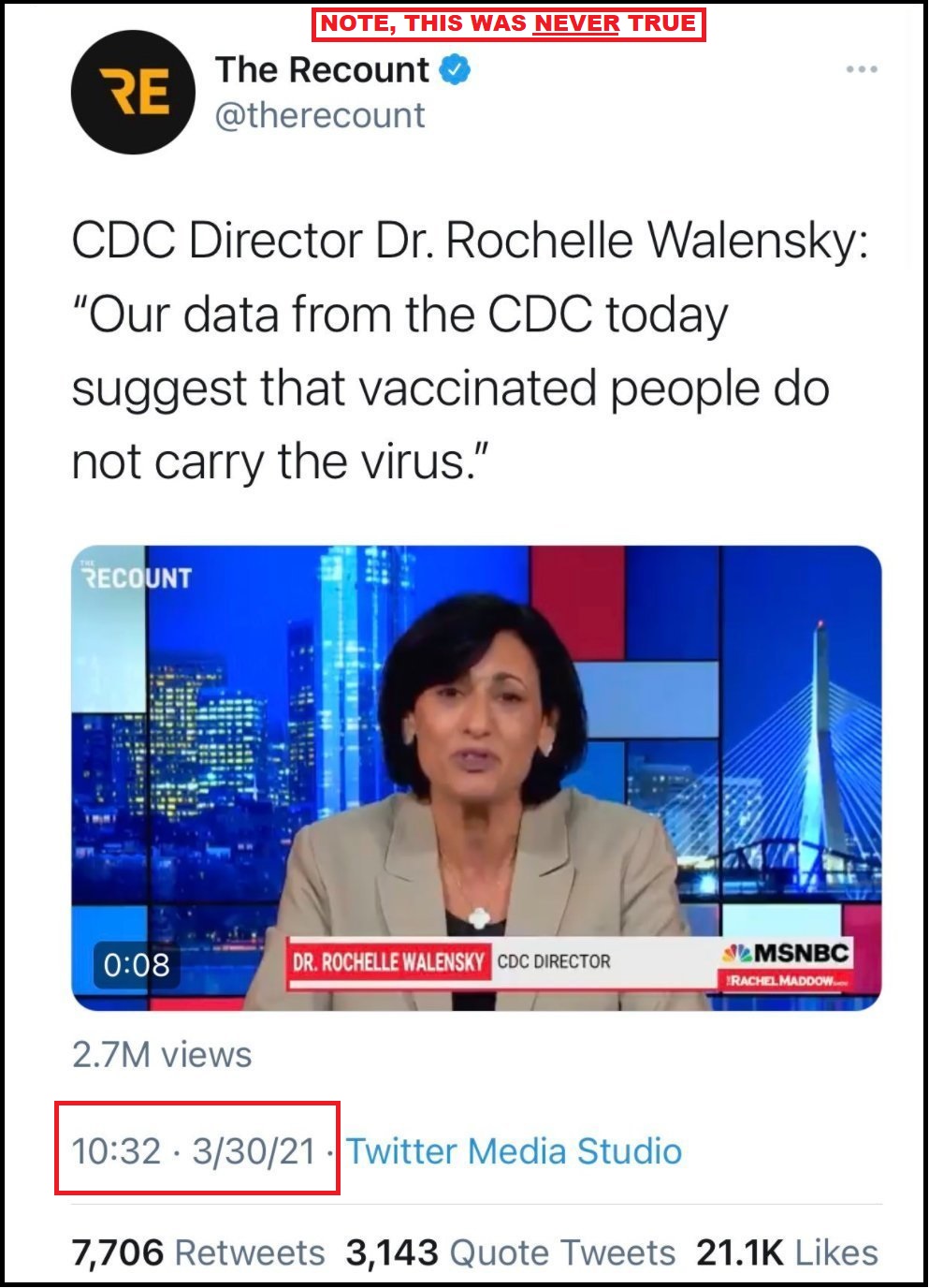
CDC Director Rochelle Walensky, a woman of notoriously sketchy flip-floppery disposition, has decreed new boosters for most.
Because the life-saving vaccine products are the greatest thing ever, the FDA advisory panel wasn’t even needed this time. You guessed it, no medical consultations, discussions or opinions were considered prior to the CDC and FDA announcement.
Yes, you read that confidence building paragraph correctly.
CDC Director – “Today, CDC expanded eligibility for an additional booster dose for certain individuals who may be at higher risk of severe outcomes from COVID-19. Boosters are safe, and people over the age of 50 can now get an additional booster 4 months after their prior dose to increase their protection further.”
“This is especially important for those 65 and older and those 50 and older with underlying medical conditions that increase their risk for severe disease from COVID-19 as they are the most likely to benefit from receiving an additional booster dose at this time. CDC, in collaboration with FDA and our public health partners, will continue to evaluate the need for additional booster doses for all Americans.” (more)
(Via NBC) […] The FDA had already authorized a fourth shot for immunocompromised individuals. On Tuesday, the FDA also cleared a fifth shot, or second booster, for that group. Previously, immunocompromised individuals were authorized to receive a three-dose primary series of a vaccine followed by a booster.
Tuesday’s decision bypasses the independent panel of experts from both the FDA and the CDC, which have issued recommendations to the agencies throughout the pandemic on whether they should clear additional shots, and if so, for which groups.
During a call with reporters, Dr. Peter Marks, the FDA’s top vaccine regulator, said officials didn’t seek the advice of the committees because they believe the risk of harm from an additional shot, including the risk of myocarditis, is low.
He said it’s possible Americans may need to get yet another dose of a Covid vaccine this fall.
That later dose, Marks said, may not be the same version of the shots currently in use — regulators may transition to a variant-specific vaccine or one that targets more than one strain. Both Pfizer and Moderna are testing a vaccine that targets the omicron variant. Moderna is also testing a shot that targets both the delta and omicron variants.
“At some point, we are going to have to realize that this is a virus that’s going to be with us and that we have to come to grips with dealing with it on a regular basis,” he said.
The share of people who’ve received their first booster remains low in the United States, with fewer than 50 percent of those who are eligible for a booster having received one, according to data from the CDC.
Officials may have a hard time persuading people to get a second booster — as Covid cases, hospitalizations and deaths remain low, some people feel a lack of urgency and have a reduced fear of the disease, experts say. (more)

