Company claims it is manufacturing Comirnaty product with new formula.

For the uninitiated:
Comirnaty is a legally distinct product from the emergency use authorization (EUA) shots, and It has never made its way to market. For months on end, no such vaccine has ever become available. Those who received the “Pfizer shot(s)” have been injected with the emergency use authorization (EUA) version of the shots. See my piece in The Dossier for more info:
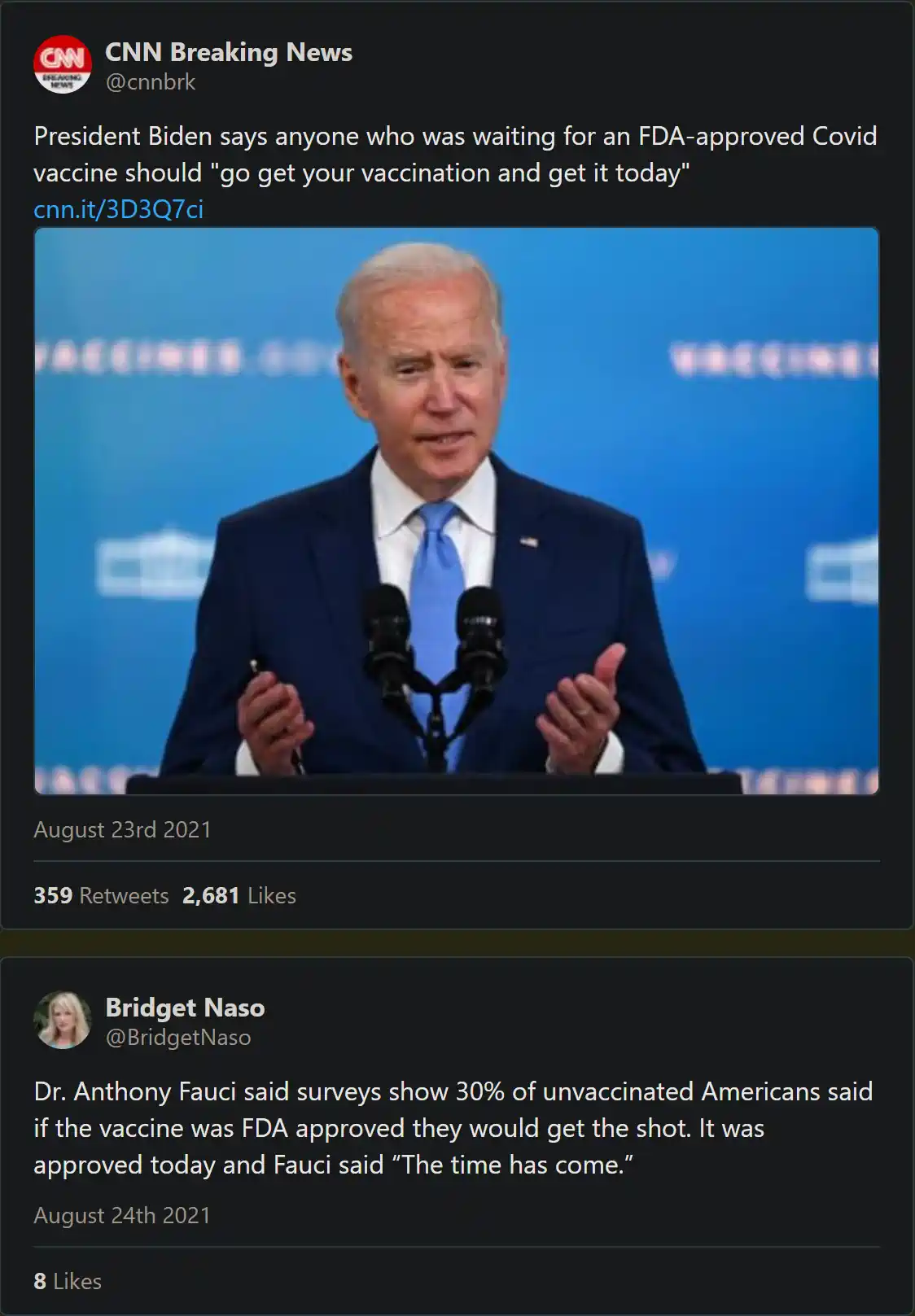
The information operation succeeded. There was indeed an FDA approved vaccine, at least on paper, but you couldn’t get it.
When originally confronted with this ordeal, Pfizer labeled this issue an inventory question that had nothing to do with the legal distinction between an experimental EUA product and an FDA-approved vaccine. Up until just weeks ago, this was the statement up on the CDC website via Pfizer:
“Pfizer received FDA BLA license on 8/23/2021 for its COVID-19 vaccine for use in individuals 16 and older (COMIRNATY). At that time, the FDA published a BLA package insert that included the approved new COVID-19 vaccine tradename COMIRNATY and listed 2 new NDCs (0069-1000-03, 0069-1000-02) and images of labels with the new tradename.
At present, Pfizer does not plan to produce any product with these new NDCs and labels over the next few months while EUA authorized product is still available and being made available for U.S. distribution. As such, the CDC, AMA, and drug compendia may not publish these new codes until Pfizer has determined when the product will be produced with the BLA labels.”
In May, Pfizer updated its statement to mention a December 2021 licensed Comirnaty product, which was granted a license four months after the highly-publicized August FDA press release.
And just last week, Pfizer finally acknowledged that its original licensed product will never be distributed. In an unreported update on the CDC website, Pfizer told the agency:
“Pfizer received initial FDA BLA license on 8/23/2021 for its COVID-19 vaccine for use in individuals 16 and older (COMIRNATY). At that time, the FDA published a BLA package insert that included the approved new COVID-19 vaccine tradename COMIRNATY and listed 2 new NDCs (0069-1000-03, 0069-1000-02) and images of labels with the new tradename. These NDCs will not be manufactured. Only NDCs for the subsequently BLA approved tris-sucrose formulation will be produced.”
The key distinction between the originally approved formulation and the tris-sucrose formulation is that — according to manufacturers — the latter can be held for a much longer period of time outside of an ultra cold freezer. These freezers cost over $10,000 a piece and each unit uses as much energy per day as an average American household. Improper storage can render the mRNA unstable.
Notably, the clinical trials for the Pfizer shot were conducted without the modified tris-sucrose ingredient. Given the partisan nature of Pfizer, the corporate media, government health bureaucracies, and your correspondent’s lack of expertise in this area, it is unclear whether this is significant.
Another notable thing to look out for in the coming days and weeks is the possibility that the subsequently FDA approved product finally becomes available in the United States. In recent days, the CDC removed the language of “not orderable at this time” above the description of both Comirnaty and Moderna’s Spikevax.
Additionally, as reported by Uncover DC, the Defense Department appears to be in the early stages of ordering what it has interpreted as a legally required minimum of Comirnaty in order to continue its mRNA mandate of American service members.